by Jack Norris, RD
Contents
- Summary
- Introduction
- PREVEND Raises Concern
- Negative Outcomes in Vitamin B12 Supplementation Trials
- Vitamin B12 and Cyanide
- Cyanocobalamin and Kidney Disease
- B12 Supplementation and Skin Rashes
- Conclusion
- Footnotes
- Bibliography
Summary
Vitamin B12 is thought to be safe in amounts much higher than the RDA. In fact, no tolerable upper intake level has been set by the Institute of Medicine. A study released in 2020 raised questions as to whether too much B12 could result in a risk for early death. This comprehensive review of the scientific literature shows that there is little reason for concern. One exception might be large doses of the cyanocobalamin form of B12 for people with chronic kidney disease.
Introduction
Vitamin B12 is required to keep blood cells and nerves healthy. B12 deficiency results in fatigue from poorly formed blood cells. It can also result in neurological problems such as tingling in the fingers and toes, the inability to walk normally, and cognitive problems such as memory loss and confusion.
The B12 molecule contains the mineral cobalt which plays a critical role in its metabolism. B12 exists in four common forms, together referred to as cobalamins due to the cobalt.
Omnivores can obtain B12 by eating animal products, although absorption and dietary intakes often decrease as people age. Because B12 isn’t naturally found in plants (A), vegans need to obtain it through fortified foods and supplements. But what if vegans are so concerned about obtaining enough B12 that they supplement with too much?
In a 1981 review, McLaren argued that typical B12 intakes in developed countries put people in a luxus state, in which there is a burden placed on the body to rid itself of B12. He argued that the typical intakes are considerably in excess of the RDAs, that total body content is far in excess of most other vitamins in relation to requirements, that amounts in the liver relate directly to intake and considerable accumulation occurs with age, and that generally accepted normal ranges in the blood are wider than most nutrients. He wondered “whether lifelong, progressive accumulation of [vitamin B12] could lead to any ill-effects through enzyme induction (McLaren, 1981).”
Nevertheless, in the interim since McLaren published his paper, other than infrequent cases of acne-like reactions to high-dose supplements (see B12 Supplementation and Skin Rashes), problems caused by B12 supplementation haven’t been observed, including in many clinical trials using high doses. It’s generally thought that because B12 is water-soluble, the body simply excretes any excess amounts (NIH, 2020). In fact, no tolerable upper intake level for B12 has been set (IOM, 1998).
PREVEND Raises Concern
In January of 2020, a report from the Prevention of Renal and Vascular End-stage Disease (PREVEND) observational study from the Netherlands correlated high plasma B12 levels with early mortality (Flores-Guerrero, 2020). This finding isn’t unusual—many studies have correlated high, or very high, B12 levels with poor health due to disease states that cause B12 levels to increase. But the results of PREVEND don’t have an obvious explanation.
The mean age of participants in PREVEND was 53.5 (SD 12.0) and included a large proportion of people with poor kidney function. B12 intake and supplementation weren’t assessed, but people who had been given vitamin B12 injections, which are prescribed to people with severe deficiency, were excluded from the study. The participants were divided into three groups based on their plasma B12 levels (C, D):
- Group 1: <251 pmol/l
- Group 2: 251-337 pmol/l
- Group 3: >337 pmol/l
After an average of 8.2 years of follow-up, Group 3 had a statistically significant, 85% increased risk of early death compared to Group 1 (fully adjusted model: HR 1.85, CI 1.16-2.97). This finding caused concern among some vegans regarding B12 supplementation.
Elevated B12 as a Marker for Disease in Observational Studies
Elevated B12 levels have long been considered a marker, but not a cause, of numerous diseases.
A large portion of the body’s storage of B12 is in the liver, and many liver diseases cause an elevation in serum B12. For example, researchers at the National Institutes of Health found, in intensive care unit patients, a strong correlation between elevated B12 levels and risk of death, but the correlation disappeared after adjusting for liver function (Callaghan, 2014). A French study found a strong association with elevated serum B12 and cirrhosis and hepatitis (Deneuville, 2009).
Various cancers can cause elevated serum B12. A Danish study found a strong decrease in short-term survival among patients with cancer whose B12 levels were 601-800 pmol/l or >800 pmol/l (Arendt, 2016). A follow-up study found similar results, in which elevated B12 was associated with a diagnosis of cancer of the upper gastrointestinal tract, liver, pancreas, lungs, or bone marrow within the first year after measurement. The association remained after five years for liver and bone marrow cancers. The authors conclude that the originally high B12 levels were indicative of undiagnosed cancer (Arendt, 2019). Numerous other cancers have been associated with an increase in B12 levels, including types of breast, colon, and stomach (Andres, 2013).
Moderately impaired kidney function can cause elevated B12 levels. The Framingham Heart Study found that elevated B12 at baseline was associated with two different markers of impaired kidney function: albuminuria and reduced glomerular filtration. But upon following up years later, a previously reported elevated B12 was not associated with a future diagnosis of either condition (McMahon, 2015). In other words, in the original analysis poor kidney function most likely caused the elevated B12 and not vice-versa.
Vitamin B12 levels can be a marker of animal product intake that, in turn, might increase the risk of some diseases. A nested case-control study from Ireland found a correlation between B12 intake and esophagus-related diseases. The researchers speculated that it could be due to a higher intake of red meat and dairy products along with a corresponding lower intake of vegetables (Sharp, 2013). Serum B12 levels weren’t measured in this study, but since serum B12 levels tend to rise in response to B12 intake, animal to plant food intake ratios should be considered in studies finding an association between serum B12 and mortality.
Elevated serum B12 can sometimes be due to immunoglobulins forming a complex with B12-transport molecules. A study found that among 1,503 elevated B12 laboratory measurements at Derriford Hospital (>667 pmol/l), 8% were due to such an immunoglobulins complex and that after these complexes were removed from the samples, B12 levels were found to be normal. Of that 8%, 71% had been previously treated with B12. It’s not clear what causes this complex and no harm from the complex has been detected (Jeffery, 2010).
Returning to PREVEND, the researchers were aware of the associations between elevated B12 and disease, and they adjusted their results for liver function, alcohol intake, and poor kidney function. They also found no association between high plasma B12 and cancer mortality (eTable 12) or cardiovascular mortality (eTable 13).
However, in PREVEND’s supplemental material, they provide an analysis excluding patients with mild to moderate loss of kidney function, defined as a glomerular filtration rate <60 mL/min/1.73 m2 (eTable 7). They also provide an analysis excluding patients with high homocysteine levels (eTable 5). In both cases, the statistical significance between elevated B12 and mortality became very weak or disappeared.
Aside from PREVEND, there have been two studies assessing the risk of death for people with higher B12 levels that included adults younger than 60 years old:
-
A 2020 report from U.S. National Health and Nutrition Examination Survey (NHANES) tracked adults (n = ~24,000, mean age = 48 ± 19) from 1999 to 2014 in multiple surveys and found no association between elevated B12 levels or B12-supplement intake and overall mortality (Wolffenbuttel, 2020).
The study was divided into categories of daily oral intake of vitamin B12-containing supplements: none, 0.1–4.9 µg, 5.0– 24.9 µg, 25.0–99 µg, 100–999 µg, and ≥1000 µg. After adjusting for age and gender, no associations were found between supplement intake and overall mortality, cardiovascular mortality, or cancer mortality.
The study population was also divided into specific serum B12 groups: low (< 140 pmol/l), possible deficiency (140-300 pmol/l), normal (300-700 pmol/l), and elevated (>700 pmol/l). The fully adjusted model was adjusted for age, gender, ethnicity, body mass index, family income, education, smoking, alcohol, glomerular filtration rate, diabetes, hypertension, cardiovascular disease, cancer, lung disease, medication use (as a proxy for other comorbidities), white blood cell count, hemoglobin, and serum folate. The adjustments were apparently made for the participants’ status at baseline (otherwise, the results are arguably over-adjusted diluting any effect of elevated B12). There was an association between B12 level < 140 pmol/l and mortality (HR 1.39, CI 1.08–1.78). There was a weak association between B12 >700 pmol/l and cardiovascular disease (HR 1.45, CI 1.01–2.06). There was no association between B12 >700 pmol/l and cancer.
The authors state, “While the findings in our study confirm the overall U-shaped association of serum B12 concentration with mortality, they lend no support to the suggestion that high serum B12 concentrations per se are harmful or detrimental.”
-
A cross-sectional survey of Australians was conducted among 2,950 people age 29-90 at baseline and deaths were measured 29 years later. Those in the highest quartile of serum B12 (≥290 pmol/l) had no increase in mortality in either the full cohort or in the cohort without cardiovascular disease at baseline. They also tested the data using a 15-year follow-up with the same result (Hung, 2003).
Another analysis on elevated B12 levels that included people younger than 60 years old studied lung cancer incidence. A 2019 meta-analysis compiled data from 5,183 case-control pairs nested within 20 prospective cohorts with a median age of 60. They found an association between elevated B12 levels and lung cancer for those with B12 levels 428 to 531 pmol/l (OR 1.16, CI 1.03 – 1.30) and >531 pmol/l (OR 1.19, CI 1.05 – 1.34). However, the results didn’t appear to be adjusted for kidney or liver function (Fanidi, 2019).
Among relatively healthy populations at baseline, in people over 60, there have been 4 prospective studies looking at a potential association between B12 levels and mortality:
-
The Newcastle 85+ study from North East England followed 766 people, aged ≥85 years without end-stage illness, for 9 years. Those with B12 levels >500 pmol/l had an increased mortality rate, compared to the group with B12 levels 148-500 pmol/l (HR 1.41, CI 1.02-1.95). For women only, each 100 pmol/l increase was associated with a 10% increased risk of mortality (HR 1.10, CI 1.04–1.16, P Mendonca, 2018).
-
A study from the United Kingdom followed 832 people, 75-84 years old at baseline, who were not residents of a nursing home or terminally ill. People were divided into tertiles based on B12 levels with the average for each being 115, 244, and 388 pmol/l. There was no difference in mortality among the three groups. Results weren’t adjusted for kidney or liver disease (Dangour, 2008).
-
A study from Spain followed 215 adults over age 60 for six years. People taking vitamin supplements at baseline were excluded. They found a trend toward higher mortality from the lowest to highest quintiles of serum B12 (≤201 pmol/l vs. >362 pmol/l, P = .043). Results weren’t adjusted for liver or kidney function (Gonzalez, 2007).
-
The Bronx Aging Study followed 440 people, aged 75 to 85 at baseline, for 6 to 9 years. There was a 10% increase in mortality with every increase of 74 pmol/l of B12. The results weren’t adjusted for kidney or liver function (Zeitlin, 1997).
There are two studies on semi-healthy populations of older people:
-
Participants (n=2,086) over age 65 (average=87.7) in the Chinese Longitudinal Healthy Longevity Survey were followed an average of 3.3 years; 45% of the participants died during that time. Participants above the 90th percentile for serum B12 levels (>540 pmol/l) had an increased risk of all-cause mortality (HR 1.30, 95% CI 1.03–1.64). When divided by sex, the trend was only significant in men (HR 1.48, 95% CI 1.03–2.13). Results were adjusted for kidney function but not liver function (Xu, 2021).
-
399 people aged ≥75 years at baseline, from the United Kingdom, were followed for an average of 69 months. There was no association between B12 levels and mortality, with the highest quintile being >325 pmol/l for men and >346 pmol/l for women. Results weren’t adjusted for kidney or liver function (Jia, 2007).
Among populations of people known or suspected to have an illness, there have been at least eleven studies measuring the relationship between elevated B12 and mortality or morbidity, typically to determine if elevated B12 is an indicator of particular diseases (Arendt, 2013; Arendt, 2016; Arendt, 2019; Callaghan, 2014; Hemmersbach-Miller, 2005; Huang, 2012; Robinson, 2011; Ryg, 2013; Salles, 2005; Sviri, 2012; Valdivia, 2020). As a whole, the studies found that while serum B12 is often highly elevated in a variety of disease states, it isn’t as specific as other diagnostic methods. These studies are not designed to determine whether excess B12 intake is unhealthy.
In summary, because so many disease states can cause B12 levels to increase, it’s hard to determine if an association of elevated B12 and mortality is due to reverse causality, especially in older populations. Many studies have found no association between elevated B12 levels and increased mortality and studies finding an association have either not adjusted for kidney and/or liver function or the associations have been statistically weak after adjustments. The studies have not measured B12 supplementation or they have excluded those supplementing with B12.
Taken as a whole, observational studies provide little evidence that raising one’s B12 levels through supplementation could cause an increased risk of mortality or chronic disease.
Side Effects and Negative Outcomes in B12 Supplementation Trials
The ideal way to examine the dangers of B12 supplementation would be to design a study, before choosing a population and taking measurements, whose primary purpose is to examine such a possibility. Unfortunately, there are no studies like this. However, there have been many clinical trials studying whether lowering homocysteine levels through folic acid, vitamin B12, and vitamin B6 supplementation can reduce cardiovascular disease events, typically in people who have a history of cardiovascular disease; some of these studies have monitored side effects.
The Cochrane Database of Systematic Reviews is a leading journal and database for systematic reviews in healthcare. In their 2017 meta-analysis of B-vitamin supplementation trials to reduce homocysteine and prevent cardiovascular events in adults at risk of or with established cardiovascular disease, they found that B-vitamin supplementation showed promise for small reductions in stroke with little benefit for other cardiovascular outcomes (Martí-Carvajal, 2017).
Regarding potential side effects, in an analysis of eight trials assessing cancer incidence, Cochrane found an almost-statistically significant increase in cancer incidence between the B-vitamin supplementation groups compared with placebo (RR 1.07, CI 1.00-1.14). The incidence of cancer was 8.27% (1,621 cases among 19,591 participants) in the placebo group versus 8.50% (1,376 cases among 16,197 participants) in the treatment group.
The B Vitamins for the Prevention of Osteoporotic Fractures (B-PROOF) trial was included in Cochrane’s meta-analysis, but they published a further follow-up (Oliai Araghi, 2019). Participants were ≥65 years old with elevated homocysteine and normal kidney function. The treatment group took 400 µg/day of folic acid plus 500 µg/day of B12 for 2 or 3 years. After 78 months of follow-up, in the analysis of only those who complied with the protocol, there was no increased risk of all cancers (HR 1.00, CI 0.99–1.00), but an increased risk of colon cancer (HR 2.17, CI 1.26–3.75). The researchers suggested the protocol may have increased the rate of tumor progression rather than initiation and hypothesized that the mechanism could be the interaction between cobalamin and folate; cyanide wasn’t mentioned. In terms of absorbed cobalamin per week, 500 µg/day of B12 is substantially more than we recommend; the only concern with our recommendations would be for slightly higher-than-normal cyanide intakes for the less-frequent-but-higher doses.
Given that B12 isn’t the only supplement in these trials, that the associations tend to be statistically weak, and that the populations were typically unhealthy, this finding isn’t enough to drive B12 recommendations, but it suggests that erring on the side of lower amounts is prudent.
Other than cancer, Cochrane combined the data from the three different clinical trials that reported other adverse events and found no increased risk among the B-vitamin groups. These included:
- B-Vitamin Atherosclerosis Intervention Trial (BVAIT) – 3-year trial of 5 mg of folic acid, 400 µg of B12, and 50 mg of B6 per day (Hodis, 2009)
- Study of the Effectiveness of Additional Reductions in Cholesterol and Homocysteine (SEARCH) – 6.7-year trial of 2 mg of folic acid and 1,000 µg of B12 per day (Armitage, 2010)
- Supplementation with Folate, Vitamin B6 and B12 and/or Omega-3 Fatty Acids (SU.FOL.OM3) – 4.7-year trial of 560 µg of 5-methyltetrahydrofolate, 20 µg of B12, and 3 mg of B6 per day (Andreeva, 2014)
Based on the results of these trials using fairly large amounts of B12, we don’t feel there is enough evidence for concern about routine vitamin B12 supplementation for vegans.
Vitamin B12 and Cyanide
One more issue regarding B12 supplementation and chronic disease is that of the cyanide in cyanocobalamin, especially as it relates to kidney disease. But we first need some general background on cyanide (skip background).
There are four forms of vitamin B12, differentiated by the side group attached to the cobalamin molecule:
- Adenosylcobalamin
- Cyanocobalamin
- Hydroxocobalamin
- Methylcobalamin
Cyanocobalamin is the form of cobalamin most commonly found in supplements and fortified foods. Hydroxocobalamin is the form usually contained in B12 shots; the hydroxyl side group has the least attraction to the cobalamin molecule. Methylcobalamin and adenosylcobalamin are the two coenzyme forms of vitamin B12, the forms of the vitamin required for the body’s chemical reactions.
Although methylcobalamin and adenosylcobalamin are the two coenzyme forms, their supplementation is not more efficient than cyanocobalamin and hydroxocobalamin because all B12 forms go through a process of being stripped of their side groups before conversion to the coenzyme forms (Obeid, 2015).
A reasonable person might wonder why a chemical as dangerous as cyanide would be part of a nutrition supplement. It just happens that cobalamin has an especially strong affinity for cyanide, making cyanocobalamin a highly stable form of B12 and therefore suitable for fortified foods and supplements.
In fact, one of the main ways to treat cyanide toxicity is through injections of hydroxocobalamin (FDA, 2018), as the cobalamin will lose the hydroxyl side group, pick up the cyanide, and then be excreted in the urine.
The cyanide molecule, when not attached to other molecules, is very toxic to animals. It works by binding to iron in the electron transport chain which prevents the cells from producing energy. However, cyanide is also found in foods as sugar-cyanide complexes known as cyanoglycosides and is ingested in small amounts on a regular basis (EFSA, 2019).
Cyanoglycosides are found in at least 2,000 plants (WHO, 2004) and in many foods such as baked goods, stone fruits and their juices, almonds, legumes, and grains (EFSA, 2019). Some pits and seeds of common fruits (e.g., apple, apricot, peach) are particularly high in cyanide (ATSDR, 2006). The cyanide can become detached from the sugar after being ingested and must be detoxified, usually via the enzyme rhodanese which converts cyanide to thiocyanate by adding a sulfur molecule. Thiocyanate is then excreted in the urine. Although most rhodanese is found in the kidney (Aminlari, 2007), the World Health Organization reports that 80% of absorbed cyanide is metabolized to thiocyanate by rhodanese in the liver (WHO, 2004).
In people with normal kidney function, the detoxification of large amounts of cyanide doesn’t appear to inhibit B12 function, as indicated by research on patients in Nigeria, hospitalized due to high cyanide intakes from diets high in cassava root. They were found to have high cyanocobalamin levels but normal B12 metabolism (IDRC, 1973).
The European Food Safety Authority (EFSA) defines an acute reference dose (ARfD) as an estimate of a substance in food or drinking water that can be ingested over a short period of time, usually during one meal or one day, without appreciable health risk to the consumer on the basis of all known facts at the time of evaluation (ECHC, 2001). The ARfD for cyanide has been set at 20 µg/kg of body weight (EFSA, 2019), which is 1,270 µg for a 140 lb (63.5 kg) person. Due to limited data, the EFSA has not established a chronic health-based guidance value (HBGV) for cyanide (EFSA, 2019).
The Centers for Disease Control and Prevention (CDC) in the United States defines a minimal risk level (MRL) as an estimate of the amount of a chemical a person can eat, drink, or breathe each day without a detectable risk to health, not including cancer (USDHHS, 2018). The CDC has set a daily MRL for oral cyanide at 50 µg/kg of body weight. The MRL seems more appropriate for our analysis because it’s targeting chronic intakes whereas the ARfD is targeting an acute intake, but because the MRL is much higher than the ARfD, to be prudent, we’ll base our analysis on the ARfD.
There are no comprehensive data on the cyanide content of diet samples in the United States (ATSDR, 2006). Canadians’ exposure to cyanide through the average diet and from the atmosphere is thought to be “extremely low” (Canada, 2018). The European Food Safety Authority provides a spreadsheet on cyanide exposure (EFSA, 2019), and our calculations showed a daily cyanide intake from food, among 37 surveys of all adults and elderly adults, to be 1.42 to 2.53 µg/kg of body weight (E).
Cyanide is found in many water supplies but in very small amounts. Data from 1988 for the Manitoba water supply showed cyanide concentrations to be below the detection limits of 1 to 10 µg/l for treated water (Canada, 2018).
As a gas, hydrogen cyanide is ubiquitous in the atmosphere (WHO, 2004). The inhalation exposure to the general U.S. non-urban, non-smoking population is estimated to be 3.8 μg/day (ATSDR, 2006).
For a 140 lb (63.5 kg) person, the average daily cyanide intake from food and air would be 113 to 164 µg per day (1.42 to 2.53 µg/kg of body weight plus 3.8 µg/day for air exposure). A 1,000 µg dose of cyanocobalamin provides an additional 19.2 µg of cyanide (F). The graph below compares those amounts to the ARfD.
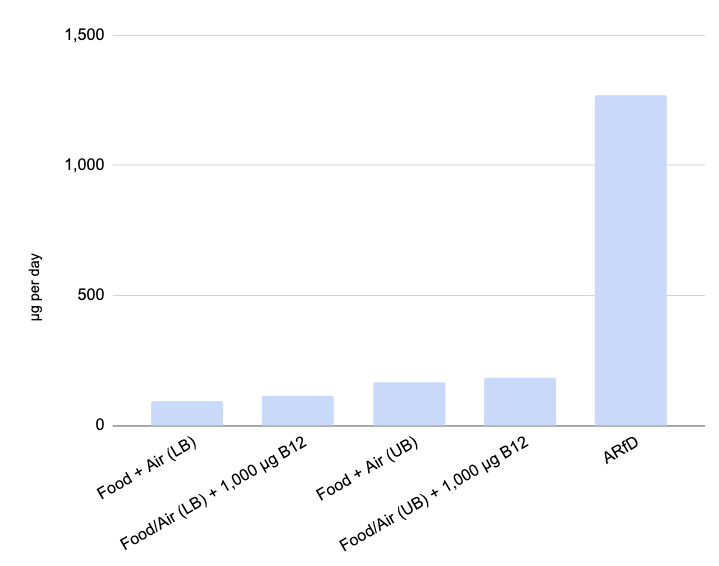
A 1,000 µg dose of cyanocobalamin increases daily cyanide intake from 12% to 20%. That seems potentially impactful, but it only increased the percentage intake of the ARfD from 7.4% to 8.9% (LB) or from 13% to 15% (UB) which is negligible.
Cyanide and the Thyroid
Another potential problem of high-dose cyanocobalamin supplementation is that thiocyanate can harm the thyroid gland through competitive inhibition with iodine. This usually occurs in geographic regions with low levels of iodine in the soil and usually, but not always, iodine supplementation reverses the disease (Amar, 2015). These problems are generally found in populations exposed to high levels of cyanide, usually through occupational sources (Dhas, 2011). We couldn’t determine the minimum amount of chronic cyanide intake that is associated with thyroid problems.
Smokers and Vitamin B12
Tobacco smoke also contains cyanide. Cyanide in smoke from U.S. commercial cigarettes ranges from 10 to 400 μg per cigarette for direct smoking and from 0.006 to 0.27 μg per cigarette for second-hand smoke (ATSDR, 2006).
The concentration of cyanide in the blood of smokers peaks immediately after smoking a cigarette, then rapidly declines with a half-life of about 4 minutes (Lundquist, 1987). The Institute of Medicine reports mixed results on whether smokers excrete more B12 than nonsmokers, but concludes that the effect of smoking on B12 requirements appears to be negligible (IOM, 1998).
Non-vegan smokers have an intake of non-cyanocobalamin forms of B12 through animal foods, while vegan smokers don’t unless they seek out a supplement. There’s no research on B12 and vegan smokers, but we’ve received no reports of any who’ve had trouble with B12 deficiency while using cyanocobalamin.
The Vitamins and Lifestyle Cohort is a prospective, observational study from the state of Washington. It found an increase in lung cancer among male smokers in the highest B12 intake group of 55–275 µg/day (Brasky, 2017). However, Obeid and Pietrzik argued, with supporting evidence, that this is likely due to reverse causality in which smokers, knowing they were at risk for health problems, were more likely to take B vitamins (Obeid, 2018).
At this time, there isn’t enough evidence to warrant separate B12 recommendations for vegan smokers, but if desired, 1,000 µg per day of methylcobalamin is an alternative option.
Cyanocobalamin and Kidney Disease
Due to the cyanide in cyanocobalamin, we recommend that people with impaired kidney function avoid cyanocobalamin supplements because of the possibility that they either don’t efficiently utilize this form of B12 or don’t efficiently clear the cyanide from their system.
The strongest evidence for the potential of cyanocobalamin to harm people with impaired kidney function comes from the Diabetic Intervention with Vitamins to Improve Nephropathy (DIVINe) clinical trial of B-vitamin supplements for lowering homocysteine levels in people with diabetic nephropathy (House, 2010). The vitamin regimen used in DIVINe was 1,000 µg of cyanocobalamin, 2.5 mg of folic acid, and 25 mg of vitamin B6 per day. After 36 months, the groups experienced the following decreases in glomerular filtration rate:
- B-vitamins: 16.5 (1.7 SE) mL/min/1.73 m2
- Placebo: 10.7 (1.7 SE) mL/min/1.73 m2
The difference between the groups of 5.8 mL/min/1.73 m2 was statistically significant (95% CI: −10.6 to −1.1; P = .02).
We charted the progression of this rate of kidney deterioration in the graph below. The blue line is roughly what would be expected in kidney decline for the average person in the age group similar to that in DIVINe (Subramanian, 2009; Shlipak, 2009). By extending the progression of decline from 36 to 60 months, the rate of kidney impairment for those taking the B vitamins would result in requiring dialysis about 15 months before the placebo group which is a 25% faster progression to kidney failure and therefore worthy of concern.
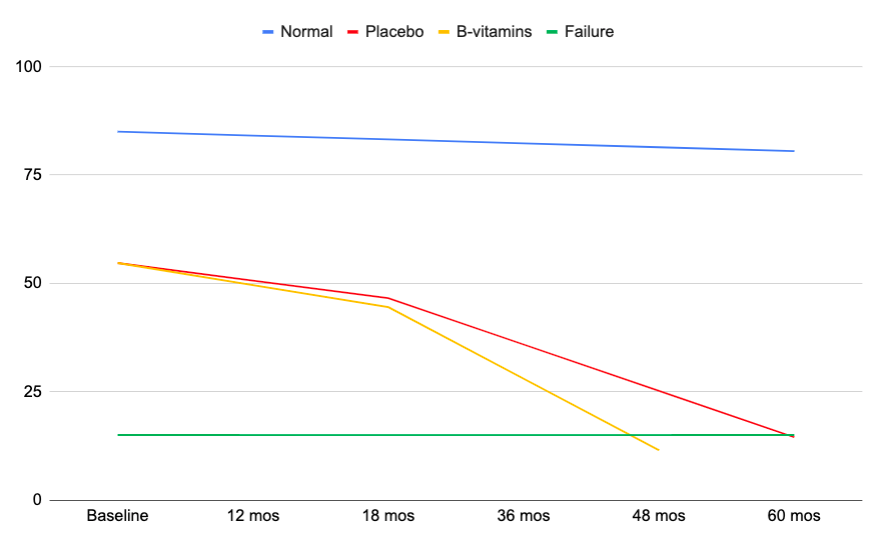
The authors had a number of hypotheses as to how these pharmacological doses of B vitamins might harm people with diabetic nephropathy, but none of the hypotheses have much evidence to support them. The authors didn’t mention any potential harm from cyanide, but in the time since, one of the DIVINe authors, David Spence, MD, suggested that the cyanide from doses of ≥400 µg/day of cyanocobalamin can be harmful to people with poor kidney function (Spence, 2019).
In contrast to the deterioration in kidney function found in the DIVINe study described above, a study on children and adolescents with diabetic nephropathy found that daily supplementation with 200 mg of thiamine (B1), 50 mg of pyridoxine (B6), and 1,000 mg of cyanocobalamin (B12) for 12 weeks significantly lowered homocysteine levels and improved kidney function as measured through microalbuminuria and cystatin C (Elbarbary, 2020). The authors speculated that these B-vitamins might be more effective at improving kidney function in the early stages of diabetic nephropathy than in the possibly-more-advanced cases in DIVINe. Elbarbary et al. also noted that their 12-week study was much shorter than the 36-month DIVINe trial.
In the section above, Vitamin B12 and Cyanide, we found that a 1,000 µg dose of cyanocobalamin increased the percentage of the ARfD for cyanide by about 1.5%, which seems negligible and unlikely to impact the progression of kidney impairment. However, we don’t believe it can be completely ruled out and until more is known, high-dose cyanocobalamin should generally not be recommended for people with kidney disease.
Spence has further thoughts on cyanocobalamin and homocysteine-lowering in patients with kidney disease (G).
B12 Supplements and Skin Rashes
Author’s note: I used AI in the literature search, analysis, and editing for this section. (Last reviewed: April 2026.)
Summary: Rare cases of people developing or exacerbating skin rashes from vitamin B12 injections have been documented. Even rarer are people who develop rashes from oral B12 supplementation. People with acne who take high-dose oral vitamin B12 should be aware that, while rare, it could possibly trigger a breakout. People with known cobalt sensitivities should be aware that vitamin B12 contains cobalt.
Sanz-Cuesta, 2020 studied the effectiveness of B12 injections compared to high-dose oral supplements and tracked side effects. Patients ≥65 years were administered 1,000 µg cyanocobalamin injections on alternate days for 2 weeks, once per week for another 6 weeks, and then once per month for about 10 months. Another group followed an oral regimen of 1,000 µg/day of cyanocobalamin for 8 weeks, followed by 1,000 µg/week for 10 months. Among the 143 receiving injections, 3 developed a rash; of the 140 taking the oral supplement, 2 developed a rash.
Kang et al. (2015, UCLA) conducted a small study providing a plausible biological explanation for why B12 supplementation may trigger acne in some individuals. The researchers found that elevated B12 levels suppress the bacterium Propionibacterium acnes‘s own B12 biosynthesis pathway, redirecting metabolic precursors toward the production of porphyrins, which induce skin inflammation. A follow-up in vitro study by the same group found that this response is strain-specific, with acne-associated P. acnes strains producing more porphyrins in response to B12 while health-associated strains did not (Johnson, 2016, UCLA).
Kang et al. monitored ten subjects who were already receiving 1,000 µg hydroxocobalamin injections for general well-being, and one developed acne within a week of an injection. Kang et al. note that a single injection can raise serum B12 to between 1,500 and 57,000 pg/mL for at least two weeks, far above the levels typically seen with oral B12. We’re not aware of research on whether more modest elevations produced by oral supplementation have similar effects on skin bacteria. Those who notice new or worsening acne after beginning B12 supplementation may find it worth trying a lower or less frequent dose before concluding supplementation is the cause.
A case series reported five women who developed acneiform eruptions from vitamin B12 therapy: three from B12 injections alone, one from combined B12 and B6 injections, and one from an oral B-vitamin combination; they had complete spontaneous remission in all cases within three to six weeks of discontinuation (Veraldi, 2018, Italy). Another case report described rosacea fulminans appearing two weeks after a teenager with pre-existing acne vulgaris and facial flushing began taking a B-complex supplement containing 20 µg of B12 and 80 mg of B6. The authors suggested that the B6, at a dose approaching the adult tolerable upper intake level, may have precipitated the eruption in a predisposed individual, making this case of limited relevance to B12 supplementation (Jansen, 2001).
Cobalt Allergies
People with a known cobalt allergy, typically identified by reactions to metal jewelry or implants, may occasionally react to vitamin B12 supplements because cobalt is a structural component of all cobalamins. Reactions, where they occur, tend to involve the skin: contact dermatitis, hives, or, in rare cases, hyperpigmentation. Though the evidence base is limited to a small number of case reports, those with known cobalt allergy are advised to discuss B12 supplementation with a clinician before starting (MHRA, 2023).
Desensitization Protocols
Lis (2025) describes desensitization protocols for patients who have developed sensitivity to B12 injections. The underlying evidence consists of a handful of case reports, but it might be helpful for clinicians.
Conclusion
The preponderance of the evidence suggests that supplementing with vitamin B12 at non-pharmacological doses doesn’t cause long-term harm to people with normal kidney function.
People with kidney disease should probably avoid cyanocobalamin supplements, especially in doses of ≥400 µg per day, and should talk to their doctor about how to obtain vitamin B12.
Last updated November 2021
Footnotes
A. When vitamin B12 is injected into the soil around a plants’ roots, some plants can absorb a portion of it, but this isn’t a practical source of B12 in the food supply. Various algae have been shown to contain vitamin B12 and its analogs, but no algae in practical amounts have been found to improve B12 status in humans. For more information, see B12 in Plant Foods.
C. PREVEND measured plasma B12 rather than the more common serum B12, but plasma and serum vitamin B12 levels have been found to be essentially the same (Sviland, 1985).
D. Sources vary for normal B12 levels, such as 133–677 pmol/l (180–914 pg/ml) (Mayo, 2020) and 200–600 pmol/l (271–813 pg/mL) (Arendt, 2016). 1 pmol/l = 1.36 pg/ml.
E. The EFSA data for cyanide intake uses a lower bound and upper bound method to account for cyanide amounts below the detection or quantification levels (EFSA, 2019, p. 16).
F. In determining what a safe level of cyanocobalamin might be, it would be helpful to know how much cyanide in a dose of cyanocobalamin is absorbed into the system. The cyanide that isn’t absorbed would be excreted in the feces, preventing harm to the kidneys.
The amount of cyanide in cyanocobalamin can be calculated as follows:
Molecular weight of cyanide = 26.02 g/mol
Percentage of cyanocobalamin as cyanide = 1.92%
Cyanide in 1,000 µg dose of cyanocobalamin = 19.2 µg
A 2010 study found that a major fraction of cyanocobalamin stays intact during the process of being absorbed and delivered to the cells (Hardlei, 2010). However, we don’t know what happens to the cyanocobalamin that isn’t absorbed. It’s likely that bacteria degrade a significant portion of cyanocobalamin (Allen, 2008; Brandt, 1977), but it’s not clear if the cyanide is liberated. To be safe, we’ll assume that all cyanide from a dose of cyanocobalamin is liberated, absorbed, and that the body must detoxify, via the kidneys, the entire 19.2 µg of cyanide from a 1,000 µg cyanocobalamin supplement.
G. Despite homocysteine levels dropping in DIVINe, the B-vitamin group had a trend toward a higher risk of cardiovascular events (HR 2.2, CI 1.0-4.6). Spence et al. later performed a meta-analysis of homocysteine-lowering trials, and found that B-vitamin supplementation is more beneficial for preventing stroke in patients with a regimen of low-dose (20 µg/day) or no cyanocobalamin (RR 0.77, CI 0.67–0.89). In contrast, patients with impaired kidney function receiving high-dose cyanocobalamin (≥400 µg) saw no benefit (RR 1.04, CI 0.84–1.27). There was no group of patients with both impaired kidney function and on a low-dose or no-cyanocobalamin regimen (Spence, 2017). Direct research is needed before drawing any conclusions.
Bibliography
Ebbing M, Bønaa KH, Nygård O, Arnesen E, Ueland PM, Nordrehaug JE, Rasmussen K, Njølstad I, Refsum H, Nilsen DW, Tverdal A, Meyer K, Vollset SE. Cancer incidence and mortality after treatment with folic acid and vitamin B12. JAMA. 2009 Nov 18;302(19):2119-26. Not cited. Analysis of two trials, NORVIT and WENBIT, that found an increased risk of cancer (mostly lung cancer) and early death from supplementation of folic acid and B12 (440 µg/day). Data were included in the 2017 Cochrane meta-analysis (Martí-Carvajal, 2017).
Erdogan MF. Thiocyanate overload and thyroid disease. Biofactors. 2003;19(3-4):107-111. Not cited.
Hsu JM, Kawin B, Minor P, Mitchell JA. Vitamin B12 Concentrations in Human Tissues. Nature. 1966;210:1264–1265. Cited by McLaren, 1981.
Test Definition: B12. Vitamin B12 Assay, S. Mayo Clinic Laboratories. Accessed August 7, 2020.
McLaren DS. The luxus vitamins–A and B12. Am J Clin Nutr. 1981;34(8):1611–1616.
Shekoohi N, Javanbakht MH, Sohrabi M, Zarei M, Mohammadi H, Djalali M. Smoking Discriminately Changes the Serum Active and Non-Active Forms of Vitamin B12. Acta Med Iran. 2017;55(6):389-394. Not cited. Authors didn’t seem to fully understand vitamin B12 metabolism.
