
by Jack Norris, RD
Contents
- Essential Information
- Additional Information
- Dietary Reference Intakes
- Daily Value
- Iron Content of Plant Foods
- Vitamin C Content of Plant Foods
- Functions of Iron
- Iron Deficiency
- Athletes
- Screening for Iron Status
- Treatment for Iron Deficiency
- Iron Absorption
- Iron Status of Vegetarians
- Iron RDA and Vegetarians
- Iron and Chronic Disease
- Hemochromatosis
- Bibliography
Essential Information
People often associate iron with red meat, so you might be surprised to know that iron is plentiful in plant foods and vegans often have higher iron intakes than meat-eaters. Obtaining enough iron from vegan foods is easy if you eat legumes (beans, peas, and lentils) and dark leafy green vegetables (such as spinach and collards). Iron is found in a range of other plant foods and many countries have foods fortified with iron. Blackstrap molasses is a type of molasses that’s high in iron.
What’s more important for meeting iron needs as a vegan is to include foods high in vitamin C with meals which increases iron absorption from other foods at that meal. For example, oatmeal with orange juice at breakfast provides iron from the oats and vitamin C from the juice. The image below shows foods high in vitamin C and what amount should be eaten to significantly increase iron absorption.

Most vegans don’t need to be too concerned about iron unless they have a history of iron deficiency. One exception is long-distance runners who menstruate, as they have a high amount of red blood cell loss. If you’re prone to iron deficiency, eat plenty of meals containing foods high in iron and vitamin C and avoid coffee and tea (which decrease iron absorption) within an hour of such meals.
Vegetarians who are pregnant or breastfeeding should also pay attention to their iron needs.
Additional Information
If someone suspects they have iron deficiency, they should be screened by a physician. A wide range of diseases can cause iron deficiency, via internal bleeding, and a physician should determine if additional testing is warranted (Pasricha). It’s also important to correct iron deficiency in order to prevent increased manganese absorption (more info: Manganese).
Serious cases of iron deficiency are treated with high-dose oral or parenteral iron which is normally effective (Pasricha) but should be administered under the supervision of a physician. Physicians sometimes recommend eating meat to vegetarian clients who have iron deficiency. Meat-eaters with anemia are normally treated with iron therapy rather than being told to eat more meat; similarly, vegetarians can be treated with supplemental iron and vitamin C.
On average, vegetarians have lower iron stores than meat-eaters, but this might be largely a function of reduced inflammation. The iron from meat has been associated with an increased risk of mortality, type 2 diabetes, cardiovascular disease, and colon cancer.
Dietary Reference Intakes
| U.S. Dietary Reference Intakes (DRI) for Iron | ||
|---|---|---|
| Age | DRI (mg) |
Upper Limit (mg) |
| 0–6 mos | 0.27 | 40 |
| 7–12 mos | 11 | 40 |
| 1–3 | 7 | 40 |
| 4–8 | 10 | 40 |
| 9–13 | 8 | 40 |
| Male 14–18 | 11 | 45 |
| Female 14–18 | 15 | 45 |
| Male 19+ | 8 | 45 |
| Female 19–50 | 18 | 45 |
| Female > 50 | 8 | 45 |
| Breastfeeding ≤ 18 | 10 | 45 |
| Breastfeeding > 18 | 9 | 45 |
| Pregnancy | 27 | 45 |
Daily Value
In the United States, iron amounts listed on a nutrition label are a percentage of the Daily Value for iron which is 18 mg/day. For example, 25% of the Daily Value = .25 x 18 mg = 4.5 mg.
Iron Content of Plant Foods
The table below shows a range of plant foods including those richest in iron.
| Iron Content of Plant Foods | |||
|---|---|---|---|
| Food | Preparation | Serving | mg |
| Grape Nuts cereal | 1/2 cup | 16 | |
| Total cereal, whole grain | 1/2 cup | 8.0 | |
| Spinach | chopped, boiled | 1 cup | 6.4 |
| Collard greens | chopped, boiled | 1 cup | 4.3 |
| Amaranth | cooked | 1 cup | 5.2 |
| Swiss chard | chopped, boiled | 1 cup | 4.0 |
| Lentils | boiled | 1/2 cup | 3.3 |
| Rice, white, enriched | cooked | 1 cup | 2.8 |
| Quinoa | cooked | 1 cup | 2.8 |
| Kidney beans | boiled | 1/2 cup | 2.6 |
| Molasses, sugar cane blackstrapA | 1 tablespoon | ≥2.5 | |
| Hemp seeds | raw | 3 tablespoons | 2.4 |
| Garbanzo beans (chickpeas) | boiled | 1/2 cup | 2.4 |
| Navy (haricot) beans | boiled | 1/2 cup | 2.2 |
| Oatmeal | cooked | 1 cup | 2.0 |
| Potato, russet | baked, w/skin | 1 medium | 1.9 |
| Black beans | cooked | 1/2 cup | 1.8 |
| Edamame | cooked | 1/2 cup | 1.8 |
| Pinto beans | boiled | 1/2 cup | 1.8 |
| Tempeh | cooked | 3 oz | 1.5 |
| Dried figs | dried, raw | 1/2 cup | 1.5 |
| Sweet potato | baked, w/skin | 1 cup | 1.4 |
| Raisins | 1/2 cup | 1.4 | |
| Almonds | roasted | 1/4 cup | 1.3 |
| Peas, green | boiled | 1/2 cup | 1.2 |
| Pistachios | dry roasted | 1/4 cup | 1.2 |
| Sunflower seeds | dry roasted | 1/4 cup | 1.2 |
| Potato, white | baked | 1 medium | 1.1 |
| Kale | chopped, boiled | 1 cup | 1.0 |
| Broccoli | chopped, boiled | 1 cup | 1.0 |
| Rice, brown | cooked | 1 cup | 1.0 |
| Soymilk | 1 cup | 1.0 – 1.5 | |
| Molasses | 1 tablespoon | 0.9 | |
| Walnuts | chopped | 1/4 cup | 0.9 |
| Tomatoes | cooked | 1/2 cup | 0.8 |
| Tofu | 3 oz | 0.8 | |
| Hummus | 2 tablespoons | 0.7 | |
| Bread, whole wheat | 1 slice | 0.7 | |
| Peanut butter | 2 tablespoons | 0.6 | |
| Rice, white, unenriched | cooked | 1 cup | 0.37 |
| Data from the USDA National Nutrient Database or food labels. A. Amount is from a range of nutrition labels. Must be blackstrap, not regular molasses, and made from sugar cane, not beet sugar. |
|||
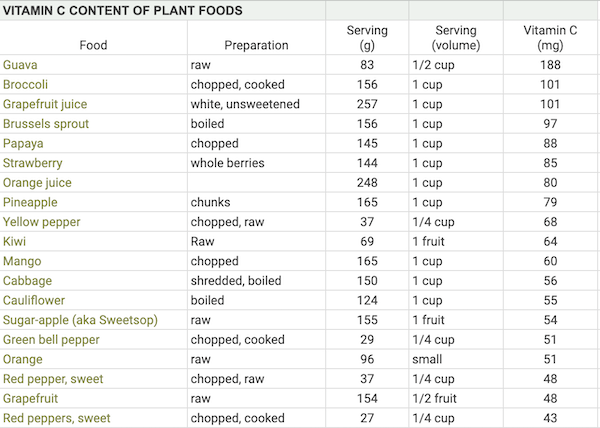
Vitamin C Content of Plant Foods
Eating foods high in vitamin C with meals increases the absorption of plant iron. The table below shows the common plant foods that are high in vitamin C. Aim for at least 50 mg of vitamin C to significantly increase iron absorption at a meal.
Our spreadsheet, Vitamin C Content of Plant Foods, list most of the plant foods highest in vitamin C.
Functions of Iron
Iron’s primary function in the body is binding oxygen. Over half of the body’s iron is part of the hemoglobin in red blood cells which transports oxygen throughout the body (Dev, 2017). Iron is also a component of myoglobin that utilizes oxygen in the heart and skeletal muscles. Iron is part of the enzyme, NADH dehydrogenase, which is part of the electron transport chain that produces ATP for energy. Iron is involved in DNA synthesis. Iron’s pro-oxidation properties are used by the immune system to destroy bacteria.
Iron Deficiency
Iron is stored on the ferritin protein and transported on the transferrin protein; both are used in the diagnosis of iron deficiency.
Pasricha et al. (2021) divide the signs and symptoms of iron deficiency into the three progressive stages of low iron stores, iron deficiency, and anemia:
- Low iron stores are marked by serum ferritin of 15–30 μg/l with a transferrin saturation of >20%. This stage of deficiency is either asymptomatic or mildly symptomatic with physical fatigue or reduced cognitive function.
- Iron deficiency is marked by a transferrin saturation of < 20% and low reticulocyte hemoglobin. It can be further divided into two types:
- Absolute iron deficiency is marked by serum ferritin of < 15-30 µg/l; it can be asymptomatic or involve fatigue, poor concentration, dizziness, tinnitus, headache, pica, or restless leg syndrome.
- Functional iron deficiency is marked by normal or increased serum ferritin (30–100 µg/l) with systemic inflammation and some of the symptoms of iron deficiency.
- Iron deficiency anemia is marked by a reduced mean cell volume (MCV) and hemoglobin concentration. The World Health Organization defines anemia as a hemoglobin level of < 120 g/l for non-pregnant females ≥15 years old, < 110 g/l in pregnancy, and < 130 g/l for males ≥15 years old.
Other consequences of iron deficiency include koilonychia (spoon-shaped nails where the outer edges are raised) , soft nails, glossitis, cheilitis (dermatitis at the corner of the mouth), mood changes, muscle weakness, and impaired immunity (EFSA), and hair loss.
The European Food Safety Authority provides a more comprehensive set of markers for iron deficiency for a variety of age groups (EFSA, Appendix B). There are many types of anemia, including B12-deficient anemia, which should be considered when determining the cause of anemia-like symptoms.
Athletes
Iron deficiency, with or without anemia, can impair muscle function and limit work capacity. Performance has been shown to improve with iron supplementation in athletes who are iron-deficient but not anemic (American Dietetic Association, 2009; Lukaski, 2004).
The average requirement for iron may be 30–70% higher for those who engage in regular, intense endurance exercise, especially running. This can be due to periods of rapid growth, training at high altitudes, menstrual blood loss, foot-strike hemolysis, intravascular hemolysis, injury, and increased losses in sweat, urine, and feces. The American College of Sports Medicine recommends that vegetarian athletes be screened regularly and aim for iron intakes above the RDA (Thomas, 2016).
Screening for Iron Status
Iron status is easily assessed through blood tests ordered by a physician. Due to concerns with both iron deficiency from menstruation and hemochromatosis (especially among men), it’s prudent to speak to a physician about having iron levels tested at least once during early adulthood. Iron deficiency can be a sign of internal bleeding.
Treatment for Iron Deficiency
Vegans with milder forms of iron deficiency should add high-iron foods and vitamin C to meals while avoiding tea, and possibly coffee, red wine, and cocoa within an hour of meals. More serious cases of iron deficiency are treated with high-dose oral or parenteral iron which is normally effective (Pasricha) but should be administered only under the supervision of a physician. Cohen and Powers (2024) provide a succinct, open-access review of treating iron deficiency anemia in adolescents.
Iron supplementation can reduce fatigue in premenopausal women (Vaucher, 2012; Verdon, 2003) and improve cognition in adolescent girls with mild iron deficiency.
The tolerable upper limit (UL) for iron is 40 mg for ages ≤ 13 and 45 mg for ages >13. The UL is based on gastrointestinal distress (Institute of Medicine, 2001). The UL is not intended for medically supervised iron therapy. Longterm iron supplementation of < 20 mg/day was not associated with an increased risk of colon cancer among women (Lee, 2004).
Iron Absorption
Iron in foods is categorized as heme and non-heme.
Heme iron comes attached to the heme molecule. Meat is the only natural source of heme iron. The average heme iron content of various cooked meats is 65% for beef, 39% for pork, and 26% for chicken and fish (Balder, 2006). Heme iron is absorbed at a rate of about 25% (EFSA, 2015). The plant-based Impossible Burger is the only plant food that contains heme iron; the heme is produced by a genetically modified yeast (Impossible Foods).
Non-heme iron absorption ranges from approximately 0.7–23% (Collings, 2013). Single-meal laboratory experiments show that absorption of non-heme iron can be reduced by phytates in legumes and whole grains and polyphenols in many herbal teas, black tea, green tea, cocoa, and red wine (Hurrell, 1999). Avoiding herbal tea within an hour of a meal can ameliorate the impact (Ahmad, 2017). Calcium supplements have also been shown to reduce iron absorption although their long-term impact on iron status appear to be minimal (Minihane, 1998; Abioye, 2021).
Siegenberg, et al. (1991) found: various doses of phytate can reduce iron absorption by 10–50%; 50 mg of vitamin C can counteract phytate; 150 mg of vitamin C can increase iron absorption to almost 30%; in the presence of a large dose of tannic acid, 100 mg of vitamin C can increase iron absorption from 2% to 8%.
A high percentage of Indian children were cured of anemia using 100 mg of vitamin C at two daily meals for 60 days (Seshadri, 1985). Researchers used 500 mg of vitamin C twice daily after meals to increase hemoglobin and serum ferritin in Indian vegetarians; vitamin C was more effective than iron supplements (Sharma, 1995).
The body can adapt to solely relying on non-heme iron. Serum ferritin is the main determinant of non-heme iron absorption: as ferritin decreases, non-heme iron absorption increases (Collings, 2013). When iron absorption decreases, ferritin excretion also decreases (Hunt, 1999).
Cooking tomato sauce in an iron skillet increased the amount of iron in the sauce and improved iron status among Brazilian teenagers and young adult lacto-ovo-vegetarians (Quintaes, 2007). The researchers considered both the acidic and water-based qualities of the tomato sauce to be important.
Beta carotene reduces the inhibitory effects of polyphenols and phytates in iron-rich grains (Garcı́a-Casal, 1998; García-Casal, 2000; Layrisse, 2000). See our chart of food high in beta carotene.
An in vitro study found that garlic and onions might increase iron absorption from grains, but this hasn’t been tested in humans (Gautam, 2010).
Iron Status of Vegetarians
Studies show that among vegetarians (including vegans), most men surpass the iron RDA of 8 mg from food while many women aged 19-50 don’t fully meet the RDA of 18 mg from food alone (Mangels, 2023).
Depending on the cutoff used, vegetarian adults and children can have a higher prevalence of iron deficiency (Haider, 2018; Gorczyca, 2013). Body mass, inflammation, and insulin resistance increase serum ferritin; adjusting for these factors ameliorated the differences between the prevalence of iron deficiency in meat-eating and vegetarian men and non-menstruating women, but not menstruating women (Slywitch, 2021).
It’s not unusual for premenopausal vegetarian women and teenage girls to have iron deficiency and sometimes anemia; it’s important that they eat iron-rich foods with a source of vitamin C at meals.
Iron RDA and Vegetarians
The Institute of Medicine (2001, p. 351) says the “requirement for iron is 1.8 times higher for vegetarians.” The Institute of Medicine bases this view on the following two clinical trials.
Hunt and Roughead (1999) performed a crossover study in which participants spent 8 weeks on a typical lacto-ovo-vegetarian diet and 8 weeks on an omnivorous diet. Iron absorption on the lacto-ovo-vegetarian diet was 1.1% compared to 3.8% on the omnivorous diet.
Cook et al. (1991) conducted a clinical trial to study iron absorption among people eating different types of meals: typical, with iron absorption enhancers, and with iron absorption inhibitors. Non-heme iron from meals was absorbed at the rates of 7.2%, 13.5%, and 2.5%, respectively. Over the course of two weeks, average absorption rates were 7.4%, 8.0%, and 3.4%, respectively. The authors say that although iron absorption from meals can vary up to 20-fold, depending on enhancers and inhibitors, population surveys haven’t demonstrated a clear relationship between their intake and iron status.
Because these trials don’t account for the body adapting its iron absorption in response to body stores, they can’t be used to determine needs for vegetarians. Longterm prospective studies on vegetarians are needed to accurately determine their iron needs but there have been none to date.
Iron and Chronic Disease
Iron is a pro-oxidant molecule. In high amounts, it could be a contributor to chronic disease. Because vegetarians have lower iron absorption and stores, their diets could be protective with respect to iron.
The association between serum ferritin levels and chronic disease is confounded by possible reverse causation due to inflammation increasing serum ferritin levels. Studying heme iron intake can be confounded by heme iron being a marker for diets higher in meat and lower in plant foods.
Regardless of the actual cause, research has associated high heme iron intakes or elevated serum ferritin with a higher risk for mortality, type 2 diabetes, cardiovascular mortality, and colon cancer. The research is detailed below.
Mortality and iron intake: A 9-year prospective analysis of the United States National Health and Nutrition Examination Study (NHANES) data found significant trends between higher heme iron intake and an increased risk of all-cause and cardiovascular mortality (Wang, 2021). A 12-year prospective analysis of NHANES II data for people age 30–70 found that iron intake >18 mg/day was associated with increased mortality, but only when combined with elevated transferrin saturation (Mainous, 2004).
Mortality and iron stores: A 12–16 year prospective analysis of NHANES II data found no relationship for mortality among white men, white women, or black men when comparing serum ferritin levels of 100-200 µg/l or >200 µg/l with 50-100 µg/l (Sempos, 2000). A 12–18 year prospective analysis of NHANES III data found no relationship between serum ferritin and mortality (Kim, 2012; Menke, 2011).
Type 2 diabetes and iron intake: A meta-analysis of prospective studies found that higher intakes of heme iron (5 studies) and iron stores (6 studies) were strongly associated with a higher risk of type 2 diabetes (Bao, 2012). There was no association for higher intakes of non-heme iron.
Type 2 diabetes and iron stores: There’s evidence that the beta cells of the pancreas, which produce insulin, are particularly susceptible to oxidation from iron due to their weak antioxidant defense mechanisms. Six male meat-eaters were given phlebotomies to reduce their serum ferritin levels and their insulin sensitivity increased (Hua, 2001).
Cardiovascular disease and iron intake: A meta-analysis of prospective studies found an increased risk of cardiovascular mortality when comparing highest versus lowest heme iron intake (RR 1.19, 95% CI 1.01–1.39) and for each 1 mg increase in heme iron intake (RR 1.25, 95% CI 1.17–1.33). There were no significant findings for non-heme or total iron intake. Some of the individual studies adjusted for dietary factors known to contribute to cardiovascular disease (Han, 2020).
Cancer and iron intake: A 9-year prospective analysis of the United States National Health and Nutrition Examination Study (NHANES) data found significant trends between higher non-heme iron intake and a lower risk for cancer mortality (Wang, 2021). A meta-analysis of 5 cohort studies found a significant and consistent, but modest, increase in the risk of colon cancer associated with heme iron intake (highest vs. lowest category: RR 1.18, 95% CI 1.06-1.32; Bastide, 2011).
Hemochromatosis
Hemochromatosis is a genetic disorder in which someone absorbs abnormally large amounts of iron, resulting in serum ferritin levels of >300 ng/ml in men, 200-300 ng/ml in postmenopausal women, and >200 ng/ml in premenopausal women (CDC, 2013). Less than 1% of people are homozygous for the hemochromatosis gene. Symptoms, especially for men, normally start to present around age 40–60 and include joint pain, fatigue, abdominal pain, and impotence. If untreated, hemochromatosis can result in liver cirrhosis, liver cancer, heart failure, and other problems.
Last updated January 2023
Bibliography
Centers for Disease Control and Prevention. Hemochromatosis (Iron Storage Disease). Accessed June 12, 2013.
Mangels R, Messina G, Messina M. The Dietitian’s Guide to Vegetarian Diets. 4th ed. Burlington, MA: Jones & Bartlett Learning; 2023.
Thomas DT, Erdman KA, Burke LM. American College of Sports Medicine Joint Position Statement. Nutrition and Athletic Performance. Med Sci Sports Exerc. 2016 Mar;48(3):543-68. This position paper expired on Dec 31, 2019. As of April 2024, it was still linked to from the list of the American College of Sports Medicine’s Position Stands for Nutrition and Athletic Performance.

12 thoughts on “Iron”
Hi! I’m a little bit confused with the iron intake.
It seems like the products listed in the table don’t really have that high iron content (other than cereals that I don’t have access to).
So as a woman in my 30s I would need to eat 2 cups of boiled spinach, 1 cup of lentils and 1 cup of oatmeal every day (just an example products), just to meet my minimum intake requirements? And each one of them with the source of vitamin C.
Do I understand it correctly or did I miss something with the numbers?
I have been diagnosed with anemia recently and am now taking the high dose ferritin supplement. But I’m trying to see how I can maintain my iron level after I’m done with my supplement, so that I wouldn’t have to supplement it forever 🙂
Thank you so much for this article and I would really appreciate your help with this question
Lena,
You’ll get some amount of iron in just about everything you eat, so you don’t need to fill the entire RDA through those foods. Once you’re off the high-dose iron supplement, you could take a multivitamin or small iron supplement to help meet the RDA. Adding a few servings of high-iron foods and vitamin C at 2-3 meals per day and meeting the RDA through a small iron supplement can improve iron status in many women.
With the iron content of plant foods table, does that take into account the absorption of the iron? so like if I wanted to use that table to reach the DRI for iron, would I need to multiply the iron content listed with my theoretical absorption factor to get the actual amount of iron I would get by eating those foods, and then use that number to see if I meet the DRI?
Max,
No, you don’t need to take into account absorption rates when determining if you’re meeting the DRI. The DRI is based on the average dietary intake needs of a population rather than on a population’s net absorption needs. But, the DRIs for iron might not exactly apply to vegans because it’s harder to absorb plant iron than the heme iron in meat (which is, on average, about half the iron in meat). Our article on Iron explains this more thoroughly.
I googled for some question and found this systematic review which seems was not mentioned in current VH article.
https://www.tandfonline.com/doi/abs/10.1080/10408398.2016.1259210?journalCode=bfsn20
I cannot access its full text, but aggregators showed me this cite:
“A few studies even reported a higher prevalence for ID and iron-deficiency anaemia (IDA) in vegetarians, especially in females ( Bhatti et al., 2007;Kajanachumpol et al., 2011;Shaw et al., 1995)”
Jack and colleagues, could you please comment on that?
Nikita,
I couldn’t find the article by Kajanachumpol, but the study by Shaw was of Buddhists in China and the study by Bhatti was of Jains in India. While these populations’ health is important, the research we present on VeganHealth, assessing the nutrition implications a vegan diet, generally focuses on vegan populations living in developed countries because we have a better idea of what their food access is like. Our recommendations would be the same for vegans in China and India, but with the hope that vegans in those countries have access to the foods or supplements we recommend or adequate alternatives.
Hi,
It would be really helpful if you could clarify the one sentence in the paragraph : Ferritin and Iron Absorption
-> When assessing studies on iron absorption, it’s important to realize that a person’s serum ferritin level is the main determinant of, and inversely proportional to, non-heme iron absorption (8).
In plain English does that mean that when a person’s blood ferritin level increases the non-heme iron absorption will decrease & the other way around (low ferritin ,higher non-heme iron absorption ) ? Thanks!
Eo.,
Yes, that’s what it means. And we point it out so that people understand, when reading the rest of the article, that non-heme iron absorption rates are always going to be relative to a study population’s iron status, so you can’t necessarily assume a specific absorption rate across all populations.
Thanks Jack,I was wondering for part 1 where you got the symptom of apathy as a result of low iron.
https://veganhealth.org/iron-part-1/#symptoms-iron-deficiency
I tried to look at the sources listed below the article (but there were some pay walls) & also found very little online about it (mainly blogs) .
Thanks!
Eo.,
I got it from one of the sources I was using when I wrote the article—I don’t know which one. Taylor Wolfram, who helps with the site, tried to find it in response to your previous comment and she said she couldn’t, so I told her to remove it from the article when she gets a chance. She’s probably planning on responding to you, but since I started responding, I figured I’d answer you about this also.
Given the other symptoms of iron deficiency, I’d be surprised if apathy isn’t also a symptom, but if Taylor couldn’t verify it, and neither can you, perhaps the source I got the info from was wrong. Or perhaps apathy is hard to measure so clinicians are unlikely to report it as much as other symptoms that are more easily detected. I’m sure there was a source that said that—I wouldn’t have made it up.
Thanks for your reply!
They state it as a symptom here: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5427299/ (but without referencing any sources)
and here (paywall) https://www.ncbi.nlm.nih.gov/pubmed/27644916
https://www.ncbi.nlm.nih.gov/pubmed/23351097 link to fulltext doesn’t work.
https://www.ncbi.nlm.nih.gov/pubmed/16108396 no link to fulltext
https://www.ncbi.nlm.nih.gov/pubmed/17066209 no link to fulltext
None of these (from what I could read) seem to point to any mechanism or discovery that apathy is a symptom of anemia. There probably is something somewhere possibly an old research paper, but I can’t seem to find it.
Currently anemic & struggling to concentrate with the fatigue, weakness, dizziness, headaches, chest pains & shortness of breath (so my research may not be so great…) Experiencing some rather large amount of apathy so I was wondering if the apathy will resolve itself or if I should be going for a psychological approach instead, that is why I was looking for the source below the article, to read more about it. It could be good to also state in the article roughly how long it takes to recover from anemia even though it varies per individual (& how bad the anemia is & the treatment method) there must be an average?
Eo.,
You should ask your doctor whether to seek psychological treatment for apathy, but given the other symptoms of iron deficiency, it wouldn’t surprise me if apathy can stem from iron deficiency. Good luck—I hope you feel better soon.
Jack