Contents
- Digestion and Absorption of Protein-Bound B12
- Digestion and Absorption of Unbound B12
- Enterohepatic Circulation
- Transport in the Blood
- Pernicious Anemia
- References
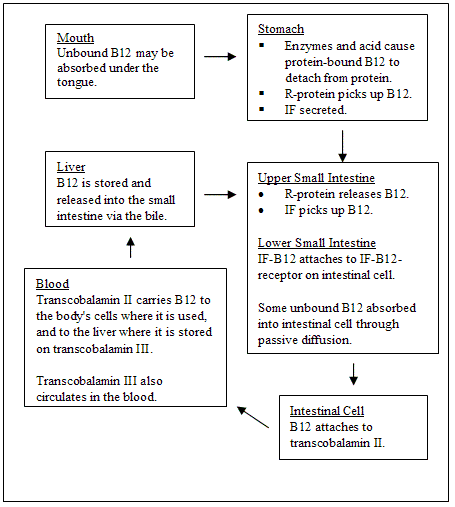
Digestion and Absorption of Protein-Bound B12
Microorganisms, primarily bacteria, are the only organisms known to manufacture B12. These bacteria are thought to live in water, soil, and the digestive tracts of animals. In animals, B12 is normally attached to a protein either for transport or storage.
When humans eat animal foods, the B12 is protein-bound. When the protein-B12 complex reaches the stomach, the stomach secretes acids and enzymes that detach the B12 from the protein. Then, in a process unique to B12, another protein, R-protein (aka cobalophilin, haptocorrin, and transcobalamin I (1)) picks up the B12 and transports it through the stomach and into the small intestine. R-protein is found in many fluids in the human body including saliva and stomach secretions. In addition to B12, R-protein can pick up any corrinoid (2).
The stomach cells also produce a protein called intrinsic factor (IF), which travels to the small intestine. When the corrinoid-R-protein complex gets to the small intestine, the corrinoid is liberated from the R-protein by enzymes made by the pancreas (3). Of the liberated corrinoids, only the cobalamins attach to intrinsic factor. Intrinsic factor then carries the cobalamins to the last section of the small intestine, the ileum.
The cells lining the ileum contain receptors for the cobalamin-IF complex. The cobalamin-IF complex protects the cobalamin against bacterial and digestive enzyme degradation (4). The IF-receptor also ensures that cobalamins will be given priority for absorption over non-cobalamin corrinoids.
In addition to the IF mechanism, passive diffusion normally accounts for 1-3% of B12 absorbed when obtained through normal food sources (3). Some inactive B12 analogues are most likely absorbed through passive diffusion.
Digestion and Absorption of Unbound B12
In supplements, B12 is not bound to protein, and therefore does not need digestive enzymes or stomach acid to be detached from a protein. Stomach acid is needed to dissolve some B12 tablets, especially if not chewed. When taken in large enough doses, unbound B12 can overcome intrinsic factor defects because so much can be absorbed through passive diffusion.
Enterohepatic Circulation
Various studies have indicated that .1-.2% of the body’s B12 pool is lost per day; the .2% loss occurs in those with pernicious anemia (5) (see below for an explanation of pernicious anemia). The average nonvegetarian stores 2,000-3,000 µg B12 (5), while losing only about 3 µg/day (6). About 60% of the total amount of B12 in the body is stored in the liver and 30% is stored in the muscles (4).
The body has a special circuit between the digestive tract and the liver. Bile, which is made in the liver and needed to digest fat, is secreted into the beginning of the small intestine. It is then reabsorbed at the end of the small intestine (the ileum) and taken back to the liver where it is used again. This circuit is called enterohepatic circulation.
People normally secrete 1.4 µg/day of B12 into their small intestines via their bile (5). Consequently, healthy people can reabsorb about .7µg B12/day from their bile (5). It is thought that in states of low B12 intake, absorption increases which can delay overt B12 deficiency, sometimes for 20-30 years (7).
For vegans who do not supplement, slight differences in enterohepatic circulation may determine how long one can go before developing B12 deficiency symptoms (8).
One study has looked at changes in serum B12 (sB12) levels in new vegans. Crane et al (9). (1994, USA) had 13 students change from a lacto-ovo vegetarian to a vegan diet:
- All 4 with sB12 in the 600-900 range fell to below 500 pg/ml in 2 months.
- 10 students followed the diet for 5 months and their average sB12 went from 417 ± 187 to 276 ± 122 pg/ml.
- After 5 months, 2 went from normal to below normal.
Transport in the Blood
After B12 is absorbed into the intestinal cells, it attaches to transcobalamin II (TC2). Transcobalamin II is made in the intestinal cells (7) where it picks up B12 and transports it to all body tissues through the blood and cerebrospinal fluid (1). Cyanocobalamin appears in the blood no longer than 5 hours after ingestion of B12 (10).
While transcobalamin II transports B12 to cells, about 3/4 of the B12 in the blood is stored on haptocorrin (aka transcobalamin I and cobalophilin) (11, 12).
Once the B12-TC2 complex arrives at the cell where it is needed, B12 is released from TC2 in the form of hydroxocobalamin. It is then turned into methylcobalamin or adenosylcobalamin (3) and used for their respective enzymes.
If the circulating B12 exceeds the binding capacity of the blood, the excess is excreted in the urine. This normally happens only after a B12 injection (5).
Pernicious Anemia
Without intrinsic factor, very little B12 is absorbed. People with intrinsic factor defects who do not get treatment eventually develop a very serious, pernicious anemia. More recently, pernicious anemia has become the term referring to people with intrinsic factor defects.
Pernicious anemia requires medical treatment. Most doctors will prescribe B12 injections, although there is evidence that oral B12 is adequate; see Oral Supplements for B12 Malabsorption.
References
1. Scalabrino G. Subacute combined degeneration one century later. The neurotrophic action of cobalamin (vitamin B12) revisited. J Neuropathol Exp Neurol. 2001 Feb;60(2):109-20.
2. Herbert V, Drivas G, Manusselis C, Mackler B, Eng J, Schwartz E. Are colon bacteria a major source of cobalamin analogues in human tissues? 24-hr human stool contains only about 5 µg of cobalamin but about 100 µg of apparent analogue (and 200 µg of folate). Trans Assoc Am Physicians. 1984;97:161-71.
3. Groff J, Gropper S. Advanced Nutrition and Human Metabolism, 3rd ed. Wadsworth: 2000.
4. Messina M, Messina V. The Dietitian’s Guide to Vegetarian Diets. Gaithersburg, MD: Aspen Publishers, Inc., 1996.
5. Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academy Press; 2000.
6. von Schenck U, Bender-Gotze C, Koletzko B. Persistence of neurological damage induced by dietary vitamin B-12 deficiency in infancy. Arch Dis Child. 1997 Aug;77(2):137-9.
7. Herbert V. Staging vitamin B-12 (cobalamin) status in vegetarians. Am J Clin Nutr. 1994 May;59(5 Suppl):1213S-1222S.
8. Donaldson MS. Metabolic vitamin B12 status on a mostly raw vegan diet with follow-up using tablets, nutritional yeast, or probiotic supplements. Ann Nutr Metab. 2000;44(5-6):229-34. And personal communication with author Jan 31, 2002.
9. Crane MG, Sample C, Patchett S, Register UD. “Vitamin B12 studies in total vegetarians (vegans). Journal of Nutritional Medicine. 1994;4:419-430.
10. Linnell JC, Matthews DM. Cobalamin metabolism and its clinical aspects. Clin Sci (Lond). 1984 Feb;66(2):113-21. Review.
11. Carmel R. Measuring and interpreting holo-transcobalamin (holo-transcobalamin II). Clin Chem. 2002 Mar;48(3):407-9.
12. Nexo E, Hoffmann-Lücke E. Holotranscobalamin, a marker of vitamin B-12 status: analytical aspects and clinical utility. Am J Clin Nutr. 2011 Jul;94(1):359S-365S. doi: 10.3945/ajcn.111.013458. Epub 2011 May 18. Review.
