by Jack Norris, RD
Table of Contents
- Summary
- Introduction
- Miscellaneous Concerns
- Limitations of Observational Research
- Fracture Rates in Context
- Body Mass Index and Caloric Intake
- Calcium
- Protein
- β-carotene and Vitamin A
- Zinc
- Selenium
- Iodine
- Vitamin D
- Vitamin B12
- Vitamin K2
- Iron
- Resistance Training
- Conclusion
- Appendices
- Appendix A: Diet Category Changes in EPIC-Oxford
- Appendix B: NSP and AOAC Fiber
- Appendix C: Censoring Due to Death
- Appendix D: Fracture Rates in the U.K. Compared to EPIC-Oxford
- Appendix E: Calcium and Vitamin D Supplements in Elderly Women with Low Vitamin D Levels
- Bibliography
Summary
A report from the EPIC-Oxford study found vegans to have a higher risk of bone fractures. Lower calcium intake accounted for about 28% of the excess risk, while lower protein intake accounted for about 9%. In this article, we examine the evidence of whether disordered eating or low intakes of β-carotene, zinc, selenium, iodine, vitamin D, vitamin B12, vitamin K2, or iron could be responsible for the remainder. Of those variables, there’s evidence for β-carotene and zinc. Vegans should pay particular attention to these nutrients (see Nutrition Tips for Vegans).
Exercise involving weights and jumping is the most reliable way for otherwise healthy people to increase their bone mineral density (see Resistance Training). I highly encourage everyone to follow a program to strengthen their bones; talk to your health professional before initiating a program if you’re injured or older.
Introduction
In November of 2020, a report from the European Prospective Investigation into Cancer and Nutrition, Oxford (EPIC-Oxford) found vegans to have a higher rate of bone fractures than meat-eaters, fish-eaters, and lacto-ovo-vegetarians (Tong, 2020).
EPIC-Oxford is conducted in the United Kingdom and is one of two large cohort studies that contain a significant portion of vegetarians and vegans; the other is the Adventist Health Study-2 from the United States and Canada.
For this particular report, EPIC-Oxford recruited participants between 1993 and 2001, and followed them until 2016. At baseline, there were 29,380 meat-eaters, 8,037 fish-eaters, 15,499 vegetarians, and 1,982 vegans. The majority of participants had followed the same diet for years before the start of the study.
About half (55.4%) of the participants completed a second follow-up questionnaire in 2010 to see if they continued to follow their original diet. If a person had changed diet groups, any subsequent bone fractures were assigned to their new diet group (more info in Appendix A: Diet Category Changes).
Over the 17.6 year follow-up period, there were 3,941 fractures including 945 hip, 889 wrist, 566 arm, 520 ankle, 366 leg, 204 vertebral, 164 rib, 99 clavicle, and 188 miscellaneous (such as in the head, hands, and feet). The researchers weren’t able to determine how many fractures were due to trauma like auto accidents and how many were due to osteoporosis.
The study found that vegans had a 43% increased risk of total fractures (from Table 2, Model 2: HR 1.43, CI 1.20-1.70). For specific fracture sites, statistically significant findings were for the hip (HR 2.31, CI 1.66-3.22) and leg (HR 2.05, CI 1.23-3.41).
When the study population was divided by sex, compared to meat-eaters, the vegan men didn’t have a significantly higher rate of total fractures (HR 1.18, CI 0.85-1.62) while the vegan women did have a significantly higher rate (HR 1.53, CI 1.24-1.88). When the study population was divided into those with a body mass index (BMI) of <22.5 and ≥22.5, vegans with a lower BMI had a significantly higher risk of total fractures (HR 1.66, CI 1.32-2.08) while vegans with a higher BMI did not (HR 1.10, CI 0.80-1.49).
There’s no question that vegans in EPIC-Oxford had a higher rate of bone fractures. But there’s a difference between vegans having a higher rate of fractures due to being vegan versus due to another reason. The rest of this article is an attempt to dispassionately examine the evidence to try to figure out what, if anything, can or needs to be done to improve the fracture rates of vegans.
Miscellaneous Concerns
There are a few concerns that we can quickly address with the information we have at this time.
Whole Foods: Some people suggested that vegans in the EPIC-Oxford cohort have generally poorer diets as shown by their lower fiber intake (28.9 g/day; Sobiecki, 2016 Table 2) compared to the fiber intake of vegans in Adventist Health Study-2 (46.7 g/day, Rizzo, 2013). Most of this discrepancy can be attributed to the different ways of measuring fiber between the two studies; if EPIC-Oxford had used the same method as Adventist Health Study-2, the vegan fiber intake would be in the range of 36.4 to 44.3 g/day. See Appendix B: NSP and AOAC Fiber for details. It’s also worth noting that in EPIC-Oxford the fiber intake for vegan women (26.4 g/day) and men (27.7 g/day) were significantly higher at baseline than for meat-eating women (18.9 g/day) and men (18.7 g/day) (Davey, 2003), suggesting that not eating enough whole plant foods is unlikely to be the explanation for the higher vegan fracture rate.
Update Feb 12, 2021: There’s a counterpoint to a lack of whole foods not playing a role in the higher fracture rates of vegans in EPIC-Oxford. β-carotene is the one nutrient for which I think there’s plausible evidence for impacting the fracture rates to a meaningful degree (see β-carotene and Vitamin A below). The RDA is 700 retinol activity equivalents (RAE) for women and 900 for men. The food highest in β-carotene is carrot juice which contains about 1,140 RAE per 1/2-cup; carrot juice may or may not fit one’s definition of a whole food. There are also processed foods such as sweet potato fries (274 RAE per cup) and pumpkin pie (about 100-150 RAE per slice) which contain fat that can aid the absorption of β-carotene. But the most common way to obtain β-carotene is through whole foods such as carrots (509 RAE per medium carrot), sweet potatoes (595 RAE per 1/2 medium), and spinach (472 RAE per 1/2-cup cooked). See Vitamin A for more foods.
Animal agriculture: A funder of EPIC-Oxford is listed as Wellcome Trust Our Planet Our Health (Livestock, Environment, and People). This is not a pro-livestock group. According to their website, “LEAP – Livestock, Environment and People – is a Wellcome funded project investigating the environmental, human health, economic and social consequences of changing patterns of global consumption of meat and dairy. The project is based at the University of Oxford and we work with a wide network of formal and informal collaborators around the world.”
Self-reporting: Some suggested the EPIC-Oxford results were unreliable because people had self-reported their diet and exercise. That could be an understandable concern of people unfamiliar with how large observational studies are conducted, but self-reporting is the only practical way to measure diet in epidemiological studies which involve tens of thousands of people. An effort is normally made to determine how valid the surveying methods are and to eliminate any self-reporting that seems suspect.
Censoring due to death: See Appendix C: Censoring Due to Death for a short explanation as to why censoring due to death probably didn’t significantly impact the results.
Limitations of Observational Research
In nutrition research, a finding with a 5% or less chance of being due to random chance is normally considered statistically significant. But that doesn’t imply causality; rather, it simply means the association probably isn’t due to random chance. This notion is reflected in the commonly repeated phrase, correlation isn’t causation.
Technically, observational research, as distinct from randomized controlled trials, never proves causation. However, for practical purposes, observational research is often used to bolster arguments for causation, and conversations about nutrition often lose sight of the fact that correlation isn’t causation.
Is there a threshold for inferring causation? Grimes and Schulz (2012) argue that relative risks in cohort studies should be less than 0.5 or greater than 2.0 for a finding to be considered worthy of interest. Harris and Zollner (2022) go further in saying, “Yet, sometimes cohort studies can infer cause-effect determinations in a situation in which it is un-ethical to apply a treatment (eg, make people smoke cigarettes or drink large amounts of alcohol long-term) and results in double-digit [10.00 or more] RR consistently across similar studies.”
For a related discussion also see Ioannidis, 2008.
Using Grimes and Schulz’s standard for inferring cause and effect would suggest that many findings regarding vegan diets, both pro and con, don’t imply causation. While this article is written using the usual 5% standard for statistical significance, we should keep in mind that many of the stastically significant associations described herein are probably not causative.
Fracture Rates in Context
What sort of difference in fracture rates does the increase found in vegans in EPIC-Oxford mean for the average person?
Based on Table 3 in Tong et al. (2020), the probability of a meat-eater suffering a fracture over the course of 10 years is 4.72%, whereas for a vegan the probability is 6.66%. If we were to observe 1,000 vegans and 1,000 meat eaters of similar age and sex over a 10-year period we would expect about 19 more vegans than meat-eaters to suffer a fracture.
How do fracture rates in EPIC-Oxford compare to the greater population of the United Kingdom? I made a crude comparison and they appeared to be roughly similar (see Appendix D: Fracture Rates in the U.K. Compared to EPIC-Oxford).
Body Mass Index and Caloric Intake
As mentioned in the introduction, right away, we know that the increased rate of fracture among vegans in this study is largely driven by women and those with a lower body mass index (BMI). This could lead us to think that a higher proportion of women with eating disorders are deciding to be vegan and that these women are more likely to have fractures.
The Centers for Disease Control and Prevention considers a BMI of 18.5 to 24.9 to indicate a “normal or healthy weight” (CDC, 2020). In EPIC-Oxford, the average BMI of vegans at baseline was 21.9 (SD 3.0) for women and 22.5 (SD 3.0) for men, compared to 24.3 (SD 4.2) and 24.9 (SD 3.3) for meat-eaters, respectively (Tong, 2020).
A 2014 meta-analysis of 25 cohort studies looking at body mass index (BMI) and bone fractures in women found that a BMI >30 is associated with a significant, modest reduction in fracture risk (Johansson, 2014). This appears to be due to a higher bone mineral density, as once bone mineral density is adjusted for the risk of fracture increases with a higher BMI. Figure 1 in the paper shows that as BMI decreases from about 22.5 to 15, the rate of fracture, especially hip fracture, increases exponentially.
So far, this research is consistent with what was found among vegans in EPIC-Oxford: Low BMI is associated with an increased risk of hip fractures.
A more recent prospective, observational study from the Nurse’s Health Study examined associations between BMI, waist circumference, and vertebral fracture risk among females with a follow-up time of about 12 years (Paik, 2019). The average BMI in the lowest quintile was 19.8 (SD 1.1) and was not associated with an increased risk of fracture.
There isn’t a lot of data on bone fractures in people with a very low BMI. A Danish study used a case-control method to follow women with eating disorders for 19.3 years (Frølich, 2020). Women with less severe cases of anorexia nervosa (n=230) had an average lowest BMI of 15.5 and a higher risk of all fractures than healthy controls (IRR 1.7, CI 1.1-2.7), but not a higher risk of hip fractures (IRR 2.9, CI 0.4–12.3). Those with a more severe case of anorexia nervosa (n=194) had an average nadir BMI of 14.4 and a much greater risk of all fractures (IRR 2.6, CI 1.8-3.7) and hip fractures (IRR 9.8, 3.6-27.7). Women with a less severe form of bulimia nervosa (n=170) had an average nadir BMI of 18.7 with no significantly elevated risk of fractures.
As with the vegans in EPIC-Oxford with a low BMI, the Danish women with severe cases of anorexia nervosa had high rates of hip fractures, but their BMI was likely much lower than the vegans in EPIC-Oxford. If the BMI for vegans is assumed to be normally distributed, then about 34% had a BMI between 18.9 and 21.9 (n=430), while about 13.5% had a BMI between 15.9 and 18.9 (n=171). The Danish women with average nadir BMIs closer to the likely BMIs of vegans in EPIC-Oxford didn’t have a higher rate of fracture.
I wrote Dr. Tammy Tong of EPIC-Oxford asking about the possibility that very low BMIs among a small percentage of vegans drove the majority of the increased risk and she responded that this was not a likely explanation, saying “For BMI and calcium, the choice of categories were driven both by the distribution of data across all dietary groups, and also after being assured that the main associations could not be driven by the extreme categories, which we checked for in preliminary analyses.”
Synthesizing the above information would suggest that although having a lower BMI was associated with a higher fracture rate in vegans, there isn’t evidence that it’s due to a high rate of disordered eating. This conclusion might be hard to follow, so I want to explain it a bit more. Women with eating disorders so severe that they result in higher rates of fracture, especially hip fracture, have BMIs on average lower than the BMIs of the vegan women in EPIC-Oxford on the lower end of the BMI spectrum (as predicted by a normal distribution). This isn’t to say that the vegans in EPIC-Oxford didn’t have a higher rate of eating disorders than the meat-eaters, but rather that even if they did have a higher rate of eating disorders, their general BMIs aren’t as low as the average BMIs in women with eating disorders who have higher rates of fracture (compared to healthy controls). So there is likely something else about being vegan (with or without an eating disorder) that is driving the higher rate of fracture among vegans.
In addition to disordered eating, a variable that would obviously be correlated with low BMI is lower caloric intake. In Table S5, EPIC-Oxford reports fracture rates adjusted for caloric intake which showed a similar total fracture rate for vegans (HR 1.42, CI 1.19-1.69) as the rate not adjusted for caloric intake from Table 2 (HR 1.30, CI 1.08-1.56). However, in the model adjusted for caloric intake, they also adjusted for BMI which would likely ameliorate any strong finding for caloric intake alone.
We can’t rule out that lower bone health could be a result of lower caloric intake, possibly leading to lower vitamin or mineral intakes among vegans.
Calcium
In EPIC-Oxford’s report (Tong, 2020), Table 2 Model 2 lists a risk of fractures for vegans compared to regular meat-eaters of 1.43 (1.20–1.70) after stratifying and adjusting for a range of variables that affect fracture risk. Model 3 uses Model 2, additionally adjusting for calcium intake, and lists a risk for vegans of 1.31 (1.10–1.57). This means that calcium intake accounts for roughly 27.9% of the excess fracture risk among vegans. The number needed to treat to prevent one fracture over 10 years with adequate calcium intake would be about 172 (2,468 / 511,459 = 0.004825; 0.004825 × 1.43 = 0.00690; 0.004825 × 1.31 = 0.006321; 0.00690 − 0.006321 = 0.000579; 1 / 0.000579 / 10 = 172.7). Adequate calcium intake might be about 1,000 mg/day, the average intake reported by regular meat-eaters in the study (including supplements), compared to an average of 591 mg/day among vegans.
However, a sub-analysis (Table 4) found that among vegans with a dietary calcium intake ≥700 mg/day, the risk of total fractures remained 1.50 (1.12–1.99). It’s worth noting that Table 4 used only dietary calcium, whereas the Table 2 Model 3 adjustment included supplemental calcium, so these findings aren’t directly comparable. Nevertheless, the Table 4 results suggest that factors beyond calcium also contribute to the elevated fracture risk among vegans, which isn’t surprising, since, based on Table 2, ~72% of the excess risk is attributable to other factors.
An earlier report from EPIC-Oxford (Appleby, 2007), found that after 5 years of follow-up, vegans had a higher fracture rate than meat-eaters (IRR 1.30, CI 1.02-1.66) that was ameliorated upon adjusting for calcium intake (IRR 1.15, CI .89-1.49). Among participants who obtained ≥525 mg/day of calcium (only 55% of the vegans compared to ~95% of the other diet groups), vegans had a similar fracture rate as meat-eaters (IRR 1.00, CI .69-1.44). The study didn’t give the average calcium intake of the vegans who obtained 525 mg/day or more, but it was possible to calculate that at a minimum, their average calcium intake was 640 mg.
Now let’s take a look at the relationship between calcium intake and fracture risk outside of EPIC-Oxford.
A meta-analysis of 44 cohort studies found that dietary calcium between about 800 to 1,000 mg per day isn’t associated with risk of fracture, and there’s no evidence that increasing dietary calcium within that range or higher prevents fractures (Bolland, 2015). (An exception is that calcium and vitamin D supplements have been found to help women aged 69 to 106 years old who normally have low calcium intake and poor vitamin D status; see Appendix E: Calcium and Vitamin D Supplements in Elderly Women with Low Vitamin D Levels).
Among omnivores with lower calcium intakes, there have been mixed results with some studies finding an increased risk of fracture among people with very low calcium intakes of about 300 mg/day or less (Xu, 2004).
A prospective, observational study of Koreans measured fracture rates of an older population with relatively low calcium intakes (Kong, 2017). Quartiles of calcium intake for women were 178, 314, 455, and 778 mg per day, and for men were 208, 344, 489, and 803 mg per day. After 9 years, there was no association between calcium intake and fracture risk for men or women.
An observational study from Sweden followed 61,433 women for 19 years (Warensjö, 2011). They were divided into quintiles of calcium intake (<751, 751-882, 882-996, 996-1,137, and >1,137). Calcium intakes below ~700 mg per day were associated with an increased risk of hip fracture, any fracture, and osteoporosis. Within the lowest quintile, the risk of fracture increased for every 100 mg decrease in calcium intake (first fracture: HR 1.08, CI 1.04-1.11; hip fracture: HR 1.07, CI 1.01-1.13). The paper provided a graph of hip fractures showing little change between calcium intakes of 700-1,000 mg/day, but a steady, exponential increase as intakes decrease from 700 to 400 mg/day.
There are concerns about supplements increasing the risk of artery calcification, although these concerns probably aren’t relevant to people on the lower end of calcium intakes (more info in Safety of Calcium Supplements).
Protein
Vegans tend to have lower protein intakes, both in absolute amounts and as a percentage of calories, and the impact of protein on bone health has been a source of controversy in the vegan community.
In EPIC-Oxford’s report (Tong, 2020), Table 2 shows that adjusting for protein intake moves the risk of fracture for vegans from 1.43 to 1.39, accounting for roughly 9.3% of the excess fracture risk among vegans. The protein adjustment was based on the percentage of calories rather than the absolute intake. Given that vegans in EPIC-Oxford had a significantly lower average caloric intake than meat-eaters, this approach may not have fully captured the effect of lower absolute protein intakes on fracture risk.
A sub-analysis (Table 4) addresses this more directly, comparing participants with dietary protein intakes ≥0.75 g/kg of body weight. Among vegans meeting this threshold, the HR for total fractures remained 1.52 (1.24–1.87). However, 0.75 g/kg is relatively low. The US RDA is 0.8 g/kg, and many researchers argue that at least 1.0 g/kg is necessary for bone and muscle health, particularly in older adults. It’s plausible that many vegans in the ≥0.75 g/kg subgroup were still getting suboptimal protein for bone health, meaning Table 4 doesn’t fully rule out protein as a contributing factor.
Now let’s take a look at the relationship between protein intake and fracture risk outside of EPIC-Oxford.
Shams-White et al. (2017) explain that early studies suggested that higher protein intakes, especially animal protein, increase urinary calcium excretion. While it was originally thought that this excreted calcium comes from the demineralization of bones, research in the 2000s showed that this calcium comes from the diet by way of increased absorption. Because protein is a major component of bone, others have speculated that increased protein can help prevent bone loss due to aging.
A meta-analysis of 20 prospective observational studies and 16 randomized controlled trials found no significant association between dietary protein and fracture risk (Shams-White, 2017). There was some evidence that more protein reduced bone loss of the lumbar spine among older adults. The study was supported by the Egg Nutrition Center and Dairy Management Inc. who were said not to have had any role in the design, analysis, interpretation, or presentation of the data or the results.
One study from France (included in the meta-analysis by Shams-White et al.) found that among women with calcium intakes lower than 400 mg/day, protein intake, which they suggested was mostly animal protein, increased the risk of fracture (Dargent-Molina, 2008).
It seems reasonable that low protein intake played some role, though likely minor, in the higher fracture rates of vegans in EPIC-Oxford.
β-carotene and Vitamin A
Preformed versions of vitamin A, known as retinoids, can be provided by animal products in the diet, whereas plant products provide the precursors to vitamin A, with the primary precursor being β-carotene. The body more tightly regulates vitamin A levels when it’s derived from precursors than when ingested as preformed vitamin A (Wu, 2014). There are a variety of theoretical ways in which too much or too little vitamin A could impact bone health (Zhang, 2017) and the research has found a variety of associations in different directions.
In their meta-analysis of 13 prospective, observational studies on vitamin A and fractures, Zhang et al. (2017) found that higher total vitamin A or retinol intake might slightly decrease the risk of total fracture but increase the risk of hip fracture. They also found that higher β-carotene intake slightly increases the risk of total fracture but not hip fracture. They observed a weak but positive influence of lower blood retinol levels on both total and hip fracture risk. They didn’t provide either intake or plasma retinol amounts, making it hard to compare with those of vegans.
Wu et al. (2014) conducted a similar meta-analysis on an almost-identical set of studies as Zhang et al. and found an increased rate of bone fracture at both the high and low ends of plasma retinol levels. They calculated the optimal blood retinol level to be 1.99 to 2.31 µmol/l. They write:
The clinical implication of this study is that blood retinol level is a double-edged sword for the risk of hip fracture. To avoid the risk of hip fracture caused by too low or too high a level of retinol concentration, we suggest that intake of beta-carotene (a provitamin A), which is mainly from plant-based foods and should be converted (through a process adjusted by a feedback mechanism) to retinol in the blood, may be better than retinol from meat (fish, liver, poultry, or dairy foods), which will be directly absorbed into the blood after intake.
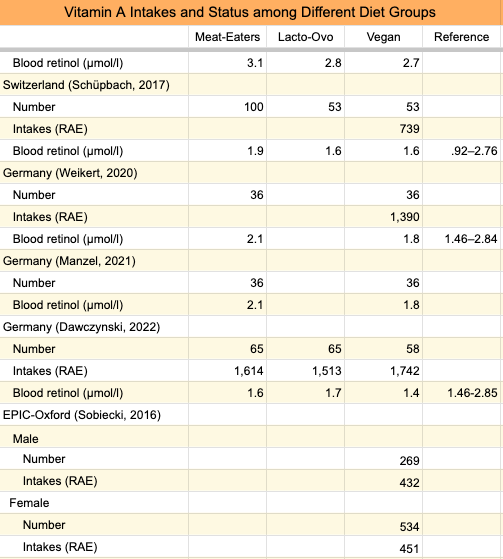
In EPIC-Oxford, after 5.2 years of follow-up, there was no association between retinol intake and bone fractures, or between β-carotene intake and fractures (Key, 2007). There was no separate analysis for vegans. The earliest nutrient intake report from EPIC-Oxford didn’t include β-carotene (Davey, 2003). A follow-up survey from EPIC-Oxford, conducted in 2010, found vegans to have β-carotene intakes of 451 RAE/day for women and 432 RAE/day for men, well below their respective DRIs of 700 RAE and 900 RAE (Sobiecki, 2016).
Of five studies measuring blood retinol levels of vegans (see the table below), one study, from Australia, found vegan levels to be slightly higher than recommended by Wu et al., while the other four studies, from Germany, found them to be lower. Note that Wu et al.’s blood retinol recommendations are for a much smaller range than what’s considered normal and that vegans fall well within the normal reference ranges listed in the table below. The range of β-carotene intakes of vegans in Li et al. and Schüpbach et al. were quite wide such that many vegans were likely not meeting the DRI.
The table shows that vegans who eat more β-carotene than those in EPIC-Oxford had retinol levels lower than what Wu et al. recommend. It, therefore, seems possible that low β-carotene intakes among EPIC-Oxford vegans could have contributed to higher fracture rates.
It’s easy for most vegans to get enough β-carotene through common, whole foods such as carrots, but if you neglect such foods it’s possible to be quite low (see Daily Needs: Vitamin A for sources and amounts). It’s best to consume food sources of β-carotene with fat for optimal absorption.
Zinc
Zinc is both a component of bone and also involved in many metabolic pathways that could impact bone health. The DRI for zinc is 11 mg for men and 9 mg for women. Vegans typically ingest close to the DRI through food, but there’s a concern that lower zinc absorption from plant foods could interfere with many vegans reaching optimal zinc status.
Studying the relationship between zinc intake and disease is complicated by the fact that zinc comes in high-protein foods and often in conjunction with a number of other minerals. There’s also no easy way to assess zinc status.
A 2021 meta-analysis of 40 studies examined associations between zinc intakes (both from food and supplements), serum zinc levels, bone mineral density and markers, and fracture risk (Ceylan, 2021). Low serum zinc levels were associated with a higher rate of osteoporosis among case-control studies. Caloric intake and zinc intake from supplements, but not from food, were associated with higher serum zinc levels. Zinc supplementation appeared to be associated with higher BMD at the femoral neck but lower BMD in the lumbar spine. Figure 6 of Ceylan et al. shows higher BMD in the lumbar spine and femur was associated with a serum zinc level of about 80 to 90 µg/dl.
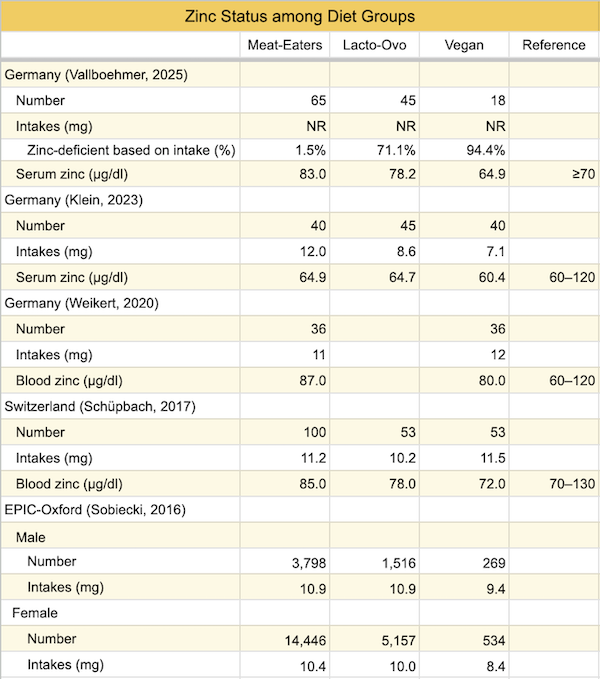
The table below lists the zinc levels and intakes of vegans compared to those of meat-eaters and lacto-ovo-vegetarians in several studies, including the EPIC-Oxford survey conducted in 2010.
The vegans in EPIC-Oxford had zinc intakes lower than the vegans in the studies from Switzerland and Germany, and the vegans from Switzerland and Germany had lower average serum zinc levels than those associated with higher bone mineral density in Ceylon et al.’s meta-analysis. One study from Germany (Vallboehmer, 2025) found a high rate of zinc deficiency among vegans. This suggests that poor zinc status might have contributed to reduced bone health among vegans in EPIC-Oxford.
Vegans should make an effort to include foods high in zinc or take a multivitamin or modest zinc supplement. See Daily Needs for recommendations.
Selenium
In a prospective study from the U.K. of 1,144 postmenopausal women, higher selenium levels were associated with greater bone mineral density at the hip, but not the lumbar spine, at study entry but not after 6 years of follow-up (Hoeg, 2012). There was also no association between selenium levels and bone fractures after 6 years of follow-up.
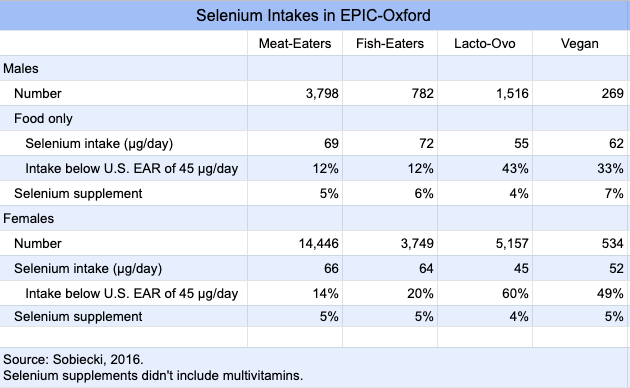
In EPIC-Oxford, a follow-up survey sent to participants an average of 14.3 years after recruitment, found that a high percentage of vegetarians weren’t meeting the estimated average requirement (EAR) for selenium (Sobiecki, 2016).
In the case of EPIC-Oxford, vegans had higher selenium intakes than lacto-ovo-vegetarians, whereas lacto-ovo-vegetarians had total fracture rates similar to meat-eaters (HR 1.07, CI 0.97-1.18). If selenium was increasing the fracture rates of vegans, it seems like it should be doing the same for lacto-ovo-vegetarians.
In contrast to selenium intakes being higher for vegans than lacto-ovo-vegetarians in EPIC-Oxford, selenium levels in toenails from the U.K. county of Norfolk showed significantly different selenium levels between omnivores (685 ng/g, n=67), lacto-ovo-vegetarians (591 ng/g, n=23), and vegans (541 ng/g, n=34). The authors reported that Norfolk’s soil is high in selenium (Judd, 1997).
While it’s unlikely that poor selenium status explains a significant portion of the higher fracture rate in EPIC-Oxford, because selenium intakes can be low in vegan diets in some countries, it’s important that vegans in those countries ensure an adequate source (see Selenium).
Iodine
Lightowler and Davies (1998) found a urinary iodine concentration (UIC) for vegans in the U.K. well below what’s considered healthy for a population, and the second-lowest average vegan UIC recorded to date. However, there are problems in using UIC to assess the iodine status of vegans (see Urinary Iodine Concentrations of Vegans). Through chemical analysis, Lightowler found baseline iodine intakes from food to be 87 µg/day which, while far short of the RDA of 150 µg/day, approximates the mean daily iodine turnover of ~95 µg/day (Zimmermann, 2012). Dietary iodine is absorbed at a rate of >92% (Zimmermann, 2012), leaving these UK vegans with a slightly lower intake, on average, than required for iodine turnover unless they were taking supplements. Key et al. (1992) found mostly normal thyroid hormone levels among vegan men from the U.K., with the notable exception being vegans who were likely receiving excessive iodine by supplementing with kelp. It’s noteworthy that vegans supplementing with kelp in Key et al. had hypothyroidism rather than hyperthyroidism.
A meta-analysis of subclinical thyroid dysfunction examined data from 17 cohort studies (Zhu, 2020). Subclinical hypothyroidism was not associated with an increased risk of any fracture (P = 0.166), hip fracture (P = 0.068), or spine fracture (P = 0.818). The only (barely) significant association between subclinical hypothyroidism and bone mineral density (BMD) was an increased BMD in the higher femur neck in women (weighted mean difference 0.04, CI 0.01-0.08, P=0.026). In contrast, subclinical hyperthyroidism (not hypo-) was associated with an increased risk of fracture (RR 1.17, CI 1.08–1.26).
Given that there isn’t an association between subclinical hypothyroidism and increased fracture rates in the general population and that while vegans in the United Kingdom might have low iodine intakes they still appear to have normal thyroid hormone levels, it seems unlikely that iodine could have played much of a role in the higher fracture rates found among vegans in EPIC-Oxford. It’s still prudent for vegans to make sure they have a reliable source of iodine (see Daily Needs).
Vitamin D
One of the more obvious nutrients to consider when assessing bone health is vitamin D, which plays a significant role in calcium homeostasis.
EPIC-Oxford didn’t measure vitamin D intakes or blood levels for all the participants and was therefore not able to adjust the fracture results for vitamin D status. However, a 2011 report from EPIC-Oxford measured the average vitamin D levels of a subset of participants. Meat-eaters (n=1,388) had an average vitamin D level of 77 nmol/l (95% CI 75-79 nmol/l) while vegans (n=89) had an average level of 56 nmol/l (95% CI 51-61 nmol/l) (Crowe, 2011).
Despite the lower vitamin D levels of vegans, their average vitamin D levels were well above the 30 nmol/l threshold determined by the Institute of Medicine (IOM) to be necessary for bone health. The IOM says, “Practically all persons are sufficient at serum 25(OH)D levels of at least 50 nmol/L,” and that optimal vitamin D levels are 50 to 125 nmol/l (Ross, 2011).
EPIC-Oxford conducted a nested case-control study after 5 years of follow-up comparing the vitamin D levels of participants who had suffered a bone fracture with those who hadn’t (Roddam, 2007). The vitamin D was measured from blood samples that had been taken at recruitment and stored. The results showed no association between vitamin D levels and risk of fracture among men or women.
Based on this evidence, it’s unlikely that lower vitamin D status played a significant role in the higher fracture rate of vegans in EPIC-Oxford.
Vitamin B12
Vitamin B12 isn’t normally thought of as a nutrient associated with bone health, but there’s evidence that it’s related.
Goerss et al. (1992) found that people with pernicious anemia (an inability to absorb vitamin B12) had a higher rate of bone fracture. In contrast, in their systematic review of studies measuring associations between poor vitamin B12 status and bone fractures, Macêdo et al (2017) found an increased risk in only 3 of 17 studies. It’s possible that a study of people with pernicious anemia could be more applicable to vegans as individuals in both groups might go extended periods without a source of B12.
Two cross-sectional studies, one from Slovakia on female lacto-ovo-vegetarians (Krivosikova, 2009) and one from Germany on male lacto-ovo-vegetarians and vegans (Herrmann, 2009), found associations between poor B12 status and markers of bone health. Although I would consider the associations fairly weak (see Vitamin B12 and Bone Mineral Density), they provide evidence that poor B12 status might play a role in fracture risk among vegans.
The average B12 levels associated with bone turnover markers in Herrmann et al. was 141 pmol/l. In EPIC-Oxford, a sub-analysis (Gilsing, 2010) found male vegans to have significantly lower serum B12 levels (122 pmol/l, n=232) than male meat-eaters (281 pmol/l, n=226) and within the range associated with poor bone health found by Herrmann et al. Vitamin B12 status of EPIC-Oxford participants has only been studied among men.
Despite male vegans in EPIC-Oxford having lower B12 status, the fracture rates were only significantly elevated among vegan women. This would suggest that B12 didn’t play a major role in the fracture rates unless vegan women had even lower B12 status than vegan men. A baseline report from EPIC-Oxford showed a similar vitamin B12 intake among vegan men (.41 µg/day) and women (.49 µg/day). While these intakes are quite a bit lower than the RDA of 2.4 µg, supplements weren’t included in the assessment (Davey, 2003) and we can’t tell from this info if B12 status would be expected to be different between male and female vegans.
One other possibility is that low B12 status impacts bone health for women more than men. While I’m not aware of any research, given that postmenopausal women suffer fractures at higher rates than men, it doesn’t seem impossible that B12 levels could be more important for women.
Of course, all vegans should ensure a reliable source of vitamin B12 regardless of its potential impact on bones (see Daily Needs).
Vitamin K2
Vitamin K comes in two main forms, K1 and K2. Vitamin K1 is found in plant foods, especially dark leafy greens, and numerous observational studies have found higher vitamin K1 intakes to be associated with a reduced risk of fractures (Hao, 2017).
While many animal products contain vitamin K2, the only plant food with appreciable amounts is the fermented soyfood natto; however, human intestinal bacteria produce vitamin K2. Vitamin K2 is touted for bone health, but in clinical trials only pharmacological doses of vitamin K2 have been used which don’t reflect dietary intakes (see Vitamin K2 and Bone Health).
An observational study from Norway found no benefit from higher intakes of vitamin K2 with respect to hip fractures, though the intakes of this population (middle quartiles ranged from 7.2 to 16.2 µg/day) were lower than the intakes in a study from the Netherlands (middle quartiles ranged from 20 to 40 µg/day), possibly explaining a lack of benefit.
At this time, there’s no evidence suggesting that a lack of vitamin K2 in vegan diets puts them at a risk for bone fractures in comparison to omnivores with usual dietary intakes.
Iron
There are many physiological mechanisms by which poor iron status could theoretically increase the risk of fracture, though there has been little observational research looking for associations. Anemia associated with thalassemia or the sickle cell trait is also associated with reduced bone health (Toxqui, 2015), but there isn’t enough data to assess whether iron status could play a role in fracture rates of vegan women.
Tong et al. (2019) measured hemoglobin levels in a large number of people in the United Kingdom, including 398 vegans. While all diet groups had average hemoglobin levels in the normal range, there was a nonsignificant trend toward lower hemoglobin among vegan men and women compared to meat-eaters. Vegan women had a non-significant trend toward higher hemoglobin levels than lacto-ovo-vegetarian women, which isn’t a surprise since vegans in EPIC-Oxford have higher iron intakes than lacto-ovo-vegetarians (Davey, 2003; Sobiecki, 2016). Given that lacto-ovo-vegetarians had a similar fracture rate to meat-eaters in EPIC-Oxford (HR 1.07, CI 0.97-1.18), it’s unlikely that the higher fracture rate of vegans is due to poor iron status.
Resistance Training
The most likely way to maintain, and often increase, bone mineral density (BMD) is by doing strength training or activities that involve jumping. This will additionally increase body mass index due to increased muscle, counteracting the potential negative impact low body mass can have on bones.
Weight-training and bouncing exercises might sound intimidating, but a home program can be done using a set of dumbbells (where weight can be changed) and a few square feet of space to step back and forth. An efficient program can take 30 minutes, three times a week, and someone can ease into it at a pace that feels energizing rather than exhausting; there are many home workout plans to get ideas from on YouTube. Warming up and easing into more weight over time is safest. Older people or those with injuries or osteoporosis should consult a healthcare professional to safely ease into a program.
Resistance Training among Vegans
Wakolbinger-Habel et al. (Austria, 2022) performed a cross-sectional study on the bone health of four groups of people aged 30 to 50: 20 vegans (9 F, 11 M) and 25 omnivores (8 F, 17 M) who performed resistance training at least once a week and 23 vegans (13 F, 10 M) and 20 omnivores (15 F, 5 M) who did no resistance training. There was little difference in bone health between vegans who performed resistance training and omnivores. Vegans who didn’t perform resistance training had poorer bone health. The duration of the vegan diet had no influence on bone microarchitecture. Vegans performing exclusively aerobic activities (n = 16) and vegans performing no sports activities at all (n = 6) had similar bone microarchitecture. The differences in bone couldn’t be explained by any obvious differences in nutrition. The researchers concluded that resistance training seems to be particularly important to preserve bone health when adhering to a plant-based diet.
Resistance Training among Omnivores
Many cross-sectional studies have found that strength athletes have higher BMD, while results are mixed for distance runners (Suominen, 1993).
A 3-year prospective study found that collegiate female basketball, volleyball, and soccer players, and track sprinters and jumpers, had high baseline bone density that continued to increase over the course of the study. In the same study, a similar pattern, though less pronounced, was found in swimmers, and thought to be due more to their complementary weight-training program than the swimming (Stanforth, 2016).
A meta-analysis of 15 studies involving multicomponent exercise (often aerobics plus resistance training) among women approximately 50 to 70 years old found that exercise regimes that combine resistance, weight-bearing exercise, and impact-aerobic activities can increase or prevent loss of muscle and skeletal mass (Marín-Cascales, 2018).
Among a small group of postmenopausal women with osteopenia and osteoporosis, Linero and Choi (2021) found that a 12-week, moderate-to-high intensity weight-training program improved markers of bone health more than did low-intensity training.
While more research is needed, increasing BMD in the upper body probably requires upper-body resistance training, and building BMD generally requires using heavier weight (Marín-Cascales et al. suggests 70-80% of someone’s 1-repetition maximum).
Yoga
Lu et al. (2016) instructed participants to do 12 minutes daily of Iyengar yoga. Of the 227 compliant participants, 83% had been diagnosed with osteoporosis or osteopenia (average age at entry was 68.2 years). After 2 years of more than every-other-day yoga, BMD increased in some locations. However, many of these participants appeared to be undergoing treatment for osteoporosis or osteopenia and there was no control group to determine if such treatment alone would have increased BMD.
Kim et al. (2015) studied the impact of 8 months of 1 hour, twice per week of Ashtanga-based yoga among premenopausal women. They found a small, positive effect on bone formation markers compared to the control group, but BMD didn’t increase.
While yoga seems promising as a method for maintaining bone, more research is needed.
Vibration Therapy
Vibration therapy shows promise in preventing and reducing osteoporosis (de Oliveira, 2023). Medical News Today says, “Whole-body vibration therapy (WBVT) may have many health benefits, including strengthening muscle and bone. This could potentially help people with osteoporosis to alleviate certain symptoms (Veazey, K. March 31, 2022).”
If you’re at risk of osteoporosis, it might be worth asking your physician about vibration therapy.
Conclusion
Given the observational nature of EPIC-Oxford, we should keep in mind that the higher bone fracture rate for vegans could be due to confounding factors. It’s also possible that a suboptimal intake of multiple nutrients, especially in people with lower body mass index, could be responsible for the higher fracture rate and that’s why there’s no single, obvious culprit.
Inadequate intake can result from a lack of information about the importance of some nutrients or from a low caloric intake. The main nutrients of concern in the EPIC-Oxford cohort are vitamin A and zinc, but vegans should pay attention to all nutrients on our Daily Needs page. Exercise involving weights and jumping is the most reliable way for otherwise healthy people to increase their bone mineral density; it also serves as a healthy way to increase body mass index and caloric intake.
Appendices
Appendix A: Diet Category Changes in EPIC-Oxford
In EPIC-Oxford, diet categories were assessed both at baseline (between 1993 and 2001) and again in 2010. A large number of people changed their diet with regard to the lacto-ovo-vegetarian and vegan categories, much of which was switching between the two (see spreadsheet). This shouldn’t have impacted the results unless there’s reason to believe someone became vegan shortly before suffering a fracture, which we currently have no reason to believe.
Appendix B: NSP and AOAC Fiber
EPIC-Oxford measured fiber as non-starch polysaccharides (NSP) and found vegan intake to average 28.9 g/day (Sobiecki, 2016). In contrast, the Adventist Health Study-2 found vegan intake to average 46.7 g/day (Rizzo, 2013). The Adventist Health Study-2 used the Nutrition Data System for Research (NDSR) 2008 database for measuring fiber in foods, which as of 2020 listed total dietary fiber as including unavailable carbohydrates (cellulose, hemicellulose, pectins, gums, and mucilages) and lignin (NDSR, 2020).
The NDSR defines fiber similarly to the Association of Official Analytical Chemists (AOAC), the definition normally used in the United States, and includes cellulose, hemicellulose, lignin, gums, modified cellulose, mucilages, oligosaccharides, pectins, and associated minor substances such as waxes, cutin, and suberin (DeVries, 1999).
The Institute of Food Science & Technology (2007) compared the fiber content of 8 foods using both the NSP and the AOAC methods; the AOAC method measured 26% more fiber than the NSP method. The UK Women’s Cohort Study compared the fiber content of different diets using both the NSP and the AOAC methods; the AOAC method measured 53% higher than the NSP method (Threapleton, 2013). Using the AOAC method in EPIC-Oxford would, therefore, show average vegan fiber intake to be closer to 36.4 to 44.3 g/day.
See the Fiber spreadsheet in the Vegan Fracture Studies for the calculations.
Appendix C: Censoring Due to Death
The EPIC-Oxford fracture results weren’t adjusted for censoring due to death (Tong, 2020). If meat-eaters died at a higher rate, or at a younger age than vegans, this could inflate the risk of fracture for vegans due to having more time in old age to fracture a bone. A previous report from the Oxford Vegetarian Study and EPIC-Oxford (Appleby, 2016) found a similar rate of death among vegetarians (including vegans) compared to meat-eaters (HR 1.02, CI 0.94-1.10); separating vegans provided a similar result (HR 1.14, CI 0.97-1.35). After excluding participants who changed diet categories during follow-up, vegetarians had a lower risk of death (HR 0.92, CI 0.84, 0.99). Dr. Tammy Tong of EPIC-Oxford doesn’t think death due to censorship could have impacted the results because there was little difference in mortality across the diet groups and also because most people in the study were still alive.
Appendix D: Fracture Rates in the U.K. Compared to EPIC-Oxford
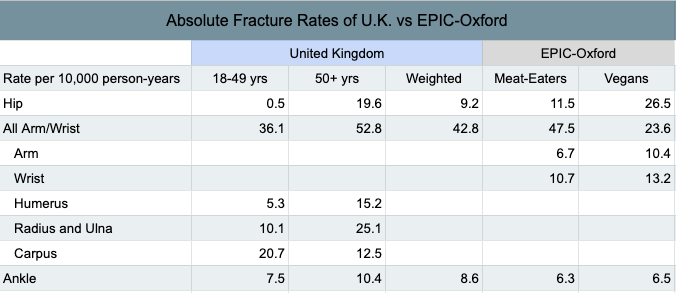
Curtis et al. (2016) published fracture rates among a representative sample of the population in the United Kingdom. It appears that they measured the total fracture rates differently than did EPIC-Oxford and so my comparison is limited to specific sites that were calculated similarly (see table below).
Curtis et al. provides separate fracture rates for ages 18 to 49 and ages 50 and older, whereas EPIC-Oxford provides rates for all ages combined. I used a crude method to weigh Curtis et al.’s rates more toward the ages of EPIC-Oxford (see table below). The rates for meat-eaters in EPIC-Oxford were similar to the overall rates in the U.K. Vegans fared much better for arm and wrist fractures combined, fared slightly better for ankle fractures, but fared much worse for hip fractures. See this spreadsheet for the calculations.
Appendix E: Calcium and Vitamin D Supplements in Elderly Women with Low Vitamin D Levels
An exception to Bolland et al.’s finding that calcium supplements don’t prevent fractures is among women age 70 and older with low calcium intakes and poor vitamin D status.
In a large clinical trial of older women (n= 3,270, average age=84 years) with low vitamin D levels, those who took 1,200 mg of calcium and 800 IU (20 µg) of vitamin D per day had significantly fewer fractures and increased bone mineral density over 18 months (Chapuy, 1992); they also had fewer fractures over a subsequent 18 months (Chapuy, 1994).
The Institute of Medicine (IOM) considers 12 ng/ml to be an adequate vitamin D level for bone health. The baseline vitamin D levels in Chapuy et al. were 16±11 ng/ml; the standard deviation of 11 ng/ml indicates that many women were well below the IOM’s recommended threshold. Furthermore, Bolland et al. say that a modern vitamin D assay would show their average vitamin D level to be 10.6 ng/ml. Calcium intake at baseline was also low at an average of 511 mg/day.
A smaller, more recent study found similar results (Chapuy, 2002).
Bolland et al. say, “On the basis of the trial data summarised here, we do not think further randomised controlled trials of calcium supplements with or without vitamin D with fracture as the endpoint in the general population are needed. In the population of frail elderly women with low dietary calcium intake and low vitamin D concentrations studied by Chapuy and colleagues, co-administered [calcium plus vitamin D] was clearly beneficial.”
Talk to your doctor about whether you might benefit from a calcium and vitamin D supplement.
Bibliography
Ho-Pham LT, Nguyen ND, Nguyen TV. Effect of vegetarian diets on bone mineral density: a Bayesian meta-analysis. Am J Clin Nutr. 2009 Oct;90(4):943-50. Meta-analysis on bone mineral density of vegetarians concluding that vegans have statistically significant, moderately lower bone mineral density than meat-eaters, but that it’s unlikely to result in a clinically important increase in fracture risk.
Institute of Food Science & Technology (IFST). Dietary Fibre. Information Statement. April, 2007. (PDF)
Johansson H, Kanis JA, Odén A, McCloskey E, et al. A meta-analysis of the association of fracture risk and body mass index in women. J Bone Miner Res. 2014 Jan;29(1):223-33. Erratum regarding author affiliation.
Kim SM, Kim AS, Ko HJ, Moon H, Choi HI, Song J. Association between Bone Mineral Density and Serum Iron Indices in Premenopausal Women in South Korea. Korean J Fam Med. 2020 May;41(3):175-182. doi: 10.4082/kjfm.18.0142. Cross-sectional. Not cited.
NDSR Nutrients, Nutrient Ratios, and Other Food Components. Nutrition Coordinating Center. University of Minnesota. 2020 version. Accessed January 30, 2021.